CHEM 112 Lecture Notes - Lecture 6: Reaction Step, Bayerischer Rundfunk, Reaction Mechanism
Document Summary
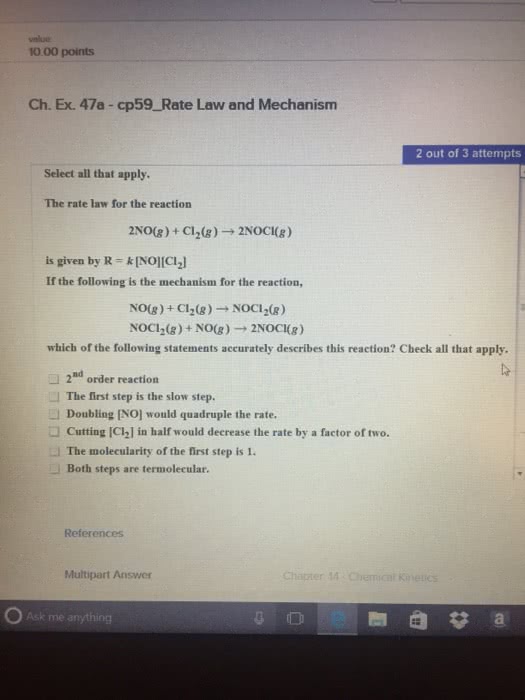
Chem112 lecture 6 more reaction mechanisms. Reaction mechanisms: describes how a reaction proceeds, can be 1 step or many elementary steps. Rate = [br2][no]2: the intermediate, or cancelling item, in this case, is br2no, each step has a rate equation and a k value, but the slow step is all that matters. Reaction mechanism and rate equations: a reaction is only fast as its slowest step, mechanisms are supported, never proven, reaction determining step it is the slow elementary step. N/a: the concentration of reactive intermediates is difficult to measure, so we have to re-write the rate law in terms of species of concentration of the information. More complexity: sometimes reaction mechanism will have intermediates within the overall rate law, or the rate determining step. In this case, you must rewrite the law, but first, find the rate law as any other. 2 h2(g) + 2 no(g) n2(g) + h2o(g) N2o2 + h2 2 n2o + h2o.