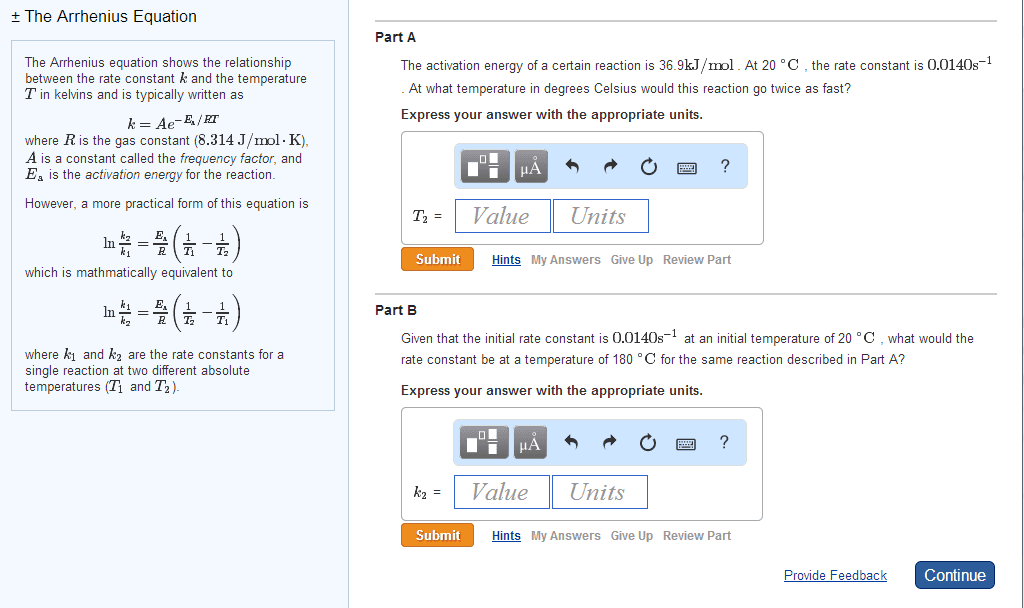
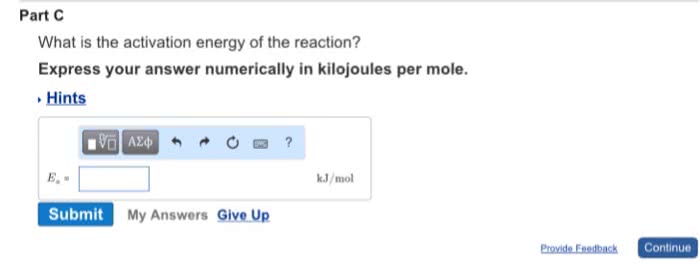
CHEM 112 Lecture Notes - Lecture 5: Rate Equation, Collision Frequency, Negative Number
Document Summary
Arrhenius equation relates k to collision frequency, energy, and temperature. Can be used to calculate activation energy or k if other values are known. A is the frequency factor, smaller value means fewer correct collisions. Ea/rt means the fraction of molecules with sufficient energy to overcome. R is the universal gas constant which is 8. 314e-3 kj/ mol. T is the temperature in kelvin so if it is in celsius just add 273 degrees. Must use the following equation ln (k2/k1)=ea/r(1/t2-1/t1) the letters for the variables still represent same numbers as above. Speeds up a reaction by lowering ea. Not consumed in a reaction but is involved. Nothing else changes besides ea large molecule and reactants have to fit just right. Reaction mechanisms refer to how a reaction proceeds. Or it could be many smaller elementary steps. Each step has a rate law and unique k value.