CHEM 112 Lecture Notes - Lecture 3: Rate Equation, List Of Association Football Teams To Have Won Four Or More Trophies In One Season, Reaction Rate
Document Summary
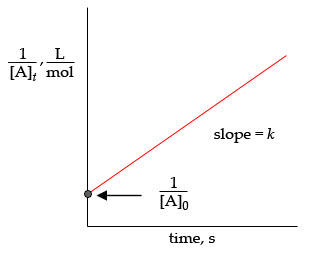
The rate constant relates reaction rate proportionally to concentration. Known as k (make sure its lowercase) Proportionally constant temperature dependent independent of concentration. Changes so that rate does not change. When concentration doubles and rate stays the same, the exponent is 0. When concentration doubles and rate doubles, the exponent is 1. When concentration doubles and rate quadruples, the exponent is 2. The sum of all exponents is the overall order of a reaction. Integrated rate law finds concentration at any point. Equation used varies based on overall order of a reaction. Half life equation: half life =1/k [a0] 0 order reactions rate does not depend on concentration. Half life equation: half life=[a0]/2k time is your x variable. Percent remaining after one half life is 50% Percent remaining after two half lives is 25% If they give you a half life but want to know information about when it has reached 25%, double the time.