CHEM 1A Lecture Notes - Lecture 17: Van Der Waals Equation, Ideal Gas Law, Ideal Gas

94
CHEM 1A Full Course Notes
Verified Note
94 documents
Document Summary
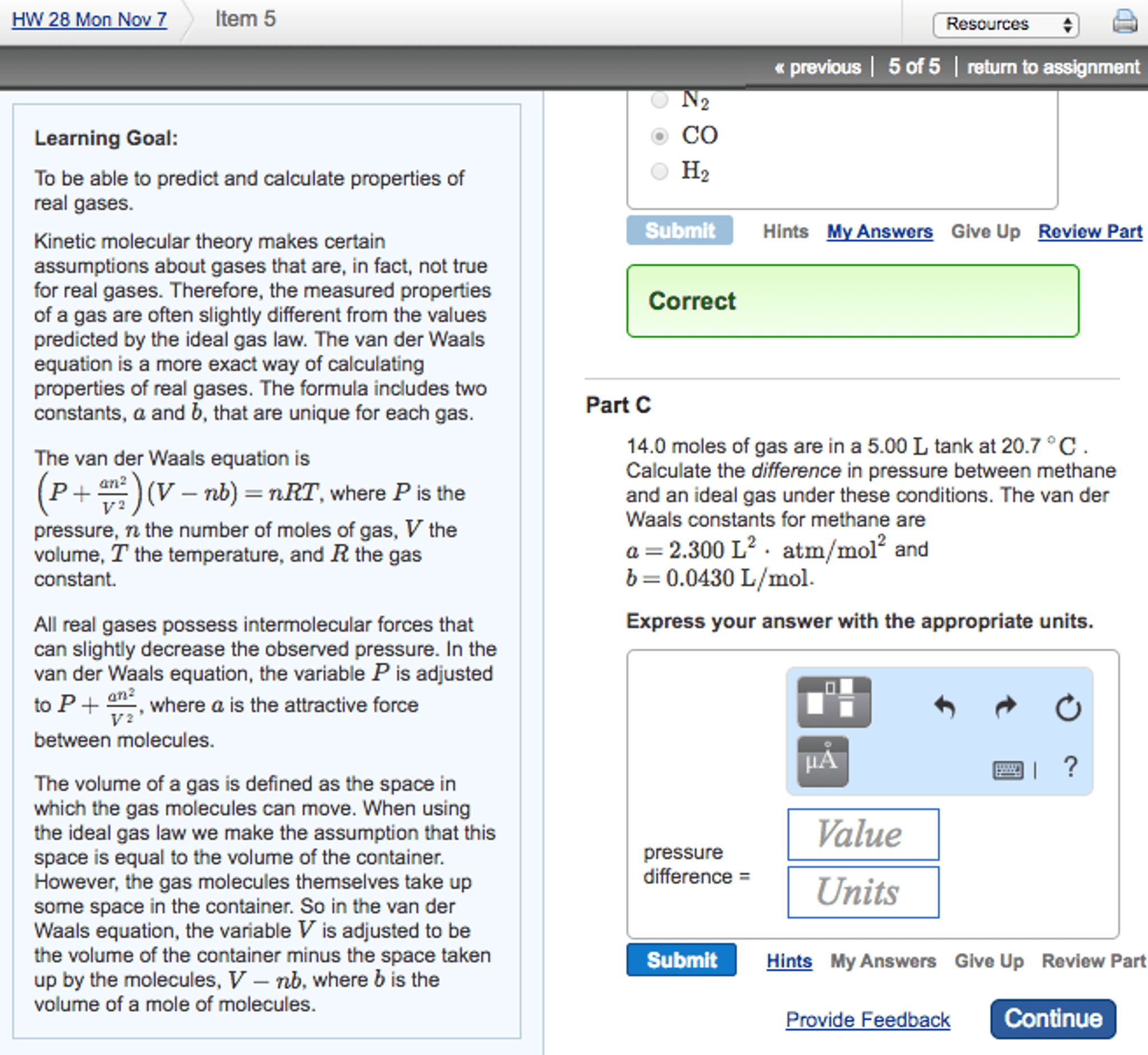
Ideal gas: real gases behave like ideal gases at: Low pressures and high temperatures: ideal gas law: pv = nrt. 1) assume volume of molecules is negligible (zero) 2) assume no force of attraction between molecules: the more you stray away from the dotted line, the less ideal the gas becomes (look at graph in lecture 17 ppt, ideal gas conditions. Low pressure = colliding less with the walls, more room to bounce around, elastic collisions remain elastic. Real gases: pabs = (nrt/v-nb) a (n/v)2, van der waals equation of state. A- term is related to intermolecular force strength. How attractive these gas molecules are to each other. The bigger the molecule is, the more attractive it is: the pressure will be decreased since the molecules are hitting the wall less. B- term is related to volume of the gas molecules. Dependent on the size of the molecule, bigger molecule is = bigger b is.