CHEM 01LB Lecture Notes - Lecture 6: Freezing-Point Depression, Melting Point, Colligative Properties

CHEM 01LB Experiment 6: Determination of Molar Mass by Freezing Point Depression
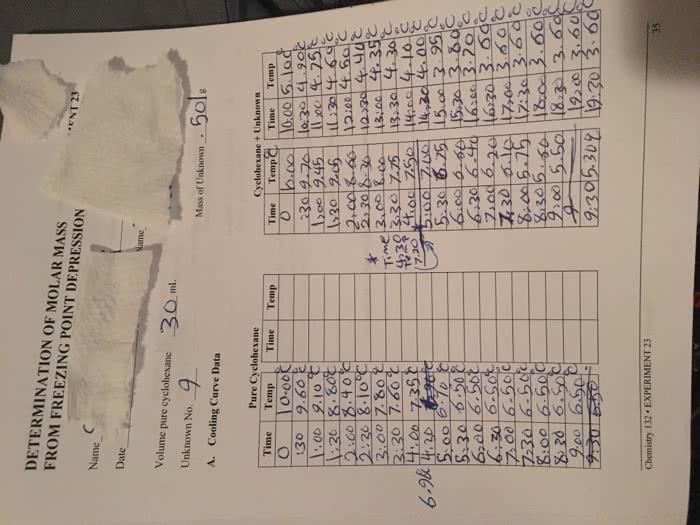
●Experimental purpose
○The purpose of this experiment is to determine the molar mass of an unknown
substance by dissolving it in a solvent with a known freezing point constant and
measuring the change in freezing point
●Freezing point depression
○Vapor pressure of a solvent is lowered when a nonvolatile substance is dissolved
in a solvent
■Freezing point is lowered
, and boiling point is raised
●Magnitude of changes depends on number of solute particles
○Colligative properties: properties of a solvent that depend only on the number of
solute particles dissolved
○Relationship between molar mass of an unknown non-dissociating solute and the
freezing point depression of a solvent:
■ΔTf = Kf m
●ΔTf = (freezing point of pure solvent) - (freezing point solution)
●Kf = freezing point constant for solvent
○Freezing point constant for lauric acid: 3.9℃/m
●m = molality
of the solute in the solvent
○Molality = kg solvent
moles solute
Document Summary
Chem 01lb experiment 6: determination of molar mass by freezing point depression. The purpose of this experiment is to determine the molar mass of an unknown substance by dissolving it in a solvent with a known freezing point constant and measuring the change in freezing point. Vapor pressure of a solvent is lowered when a nonvolatile substance is dissolved in a solvent. Freezing point is lowered , and boiling point is raised. Magnitude of changes depends on number of solute particles. Colligative properties : properties of a solvent that depend only on the number of solute particles dissolved. Relationship between molar mass of an unknown non-dissociating solute and the freezing point depression of a solvent: T f = (freezing point of pure solvent) - (freezing point solution) K f = freezing point constant for solvent. Freezing point constant for lauric acid: 3. 9 /m. M = molality of the solute in the solvent.