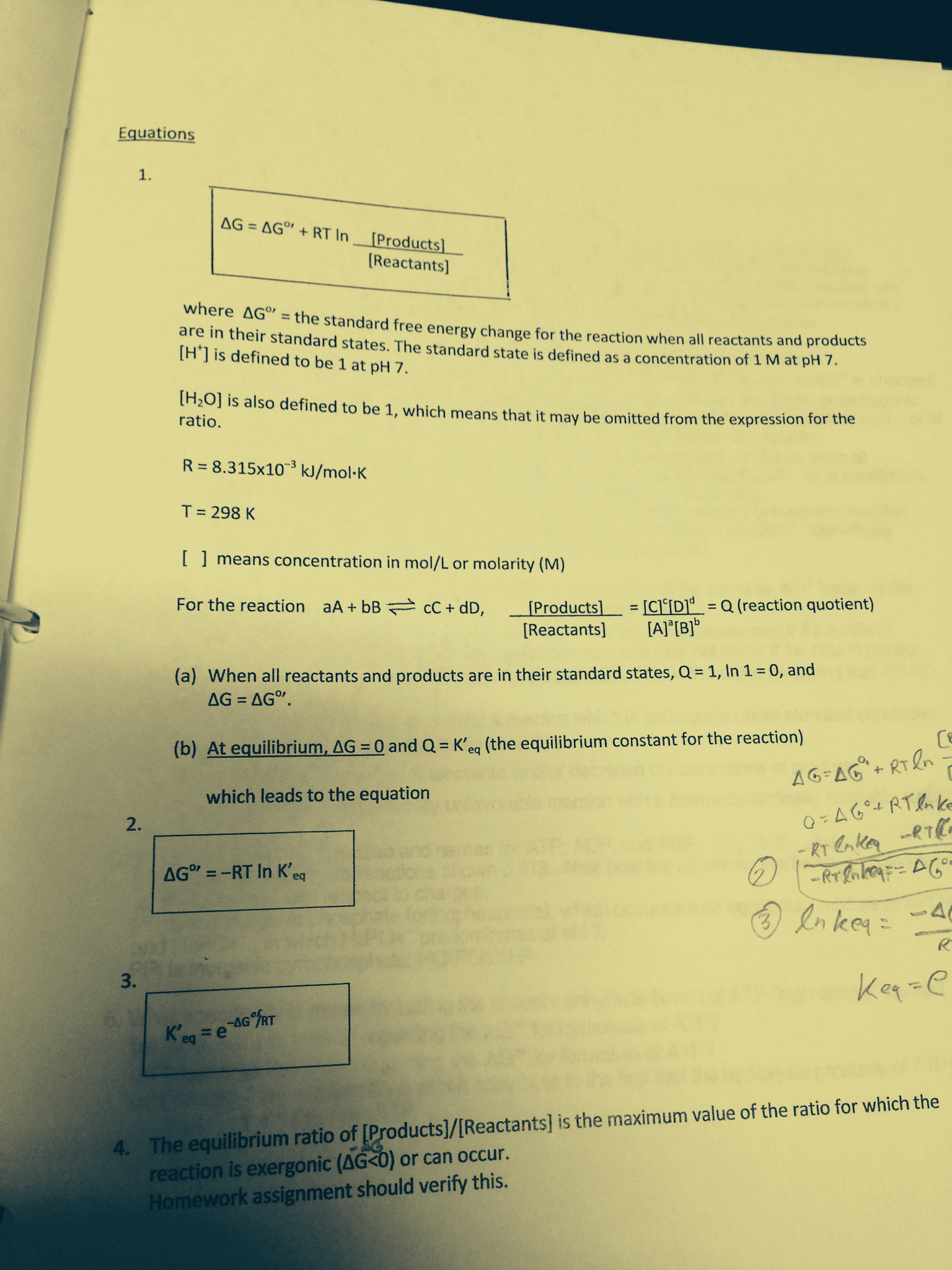CHEM 1C Lecture Notes - Lecture 2: Equilibrium Constant, Partial Pressure
13 views1 pages
Document Summary
Irreversible: 100 reactant/0 product " 0 reactant/100 product. The equilibrium constant (k): defined as a ratio (at equilibrium) where the concentration of products is raised to the concentration of reactants. If you see an equilibrium constant of 1 then the ratio between reactants and products is 50/50. If you see an equilibrium constant less than 1 then the ration between reactants and products is that reactants are greater than products. If you see an equilibrium constant of greater than 1 then the ratio between reactants and products is that products are greater than reactants. K describes the equilibrium composition of the rxn mixture. At a given temperature, k is constant. Homogeneous equilibria: where the reactants and products are all of the same phase. Heterogeneous equilibria: involves pure solids pure liquids or solvents as reactants or products, the pure solid, liquid, or solvent is not included in the equilibrium expression. We can only include gaseous and aqueous solutions.
Get access
Grade+
$40 USD/m
Billed monthly

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
10 Verified Answers
Class+
$30 USD/m
Billed monthly

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
7 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232