CHEM 1B Lecture 3: Chem 1B Smith lecture notes 3
Document Summary
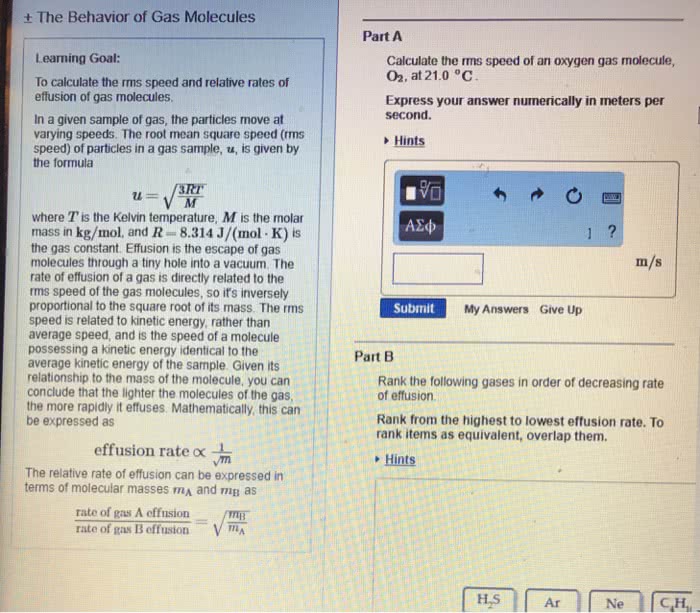
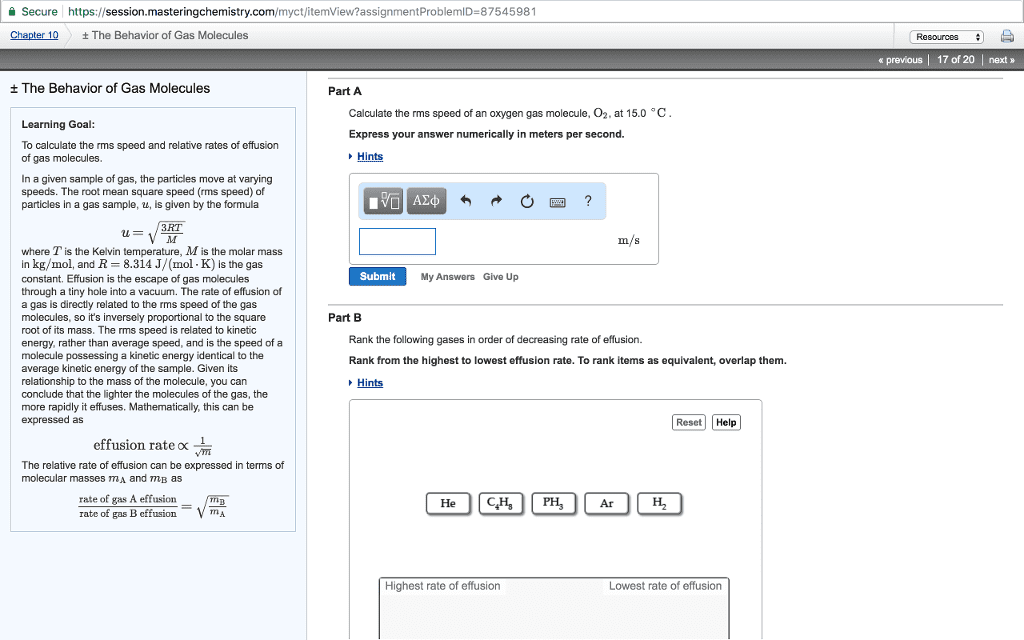
Average kinetic energy of gas molecules decreases with decreasing temp; as well as average speed. All the gas molecules in the sample cannot have the same kinetic energy. There are gas molecules that move slower than average. 1 liter of h2 liters nh3. Gas is modeled as a collection of small particles in a large volume with lots of empty space between the particles. Particles are constantly moving in random directions and with different speed. Particles undergo elastic collisions and exchange energy; there is no overall loss of kinetic energy. Temperature is defined as a measure of the average kinetic energy of particles. Root mean square speed ( m = molecular weight) Example: calculate the root mean square velocity of oxygen molecules of 25 c. Molecules do not have the same speed; molecular speed distribution can be measured experimentally. The speed distribution spreads out as temperature gets higher.