CHE 2B Lecture Notes - Lecture 9: Le Chatelier'S Principle, Reaction Quotient, Reduced Properties

64
CHE 2B Full Course Notes
Verified Note
64 documents
Document Summary
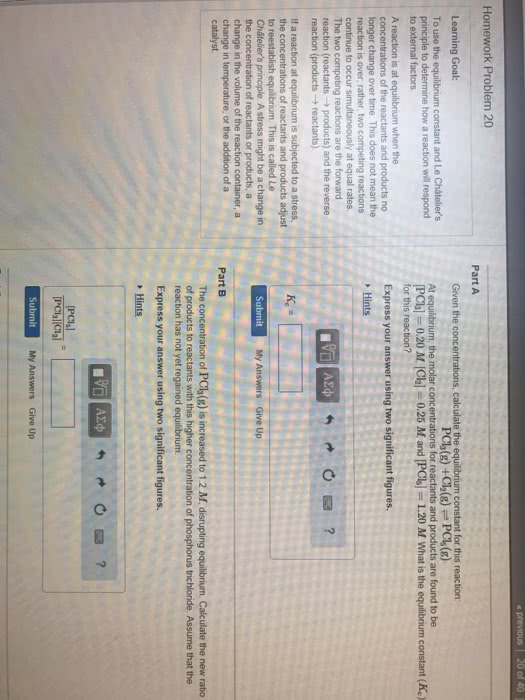
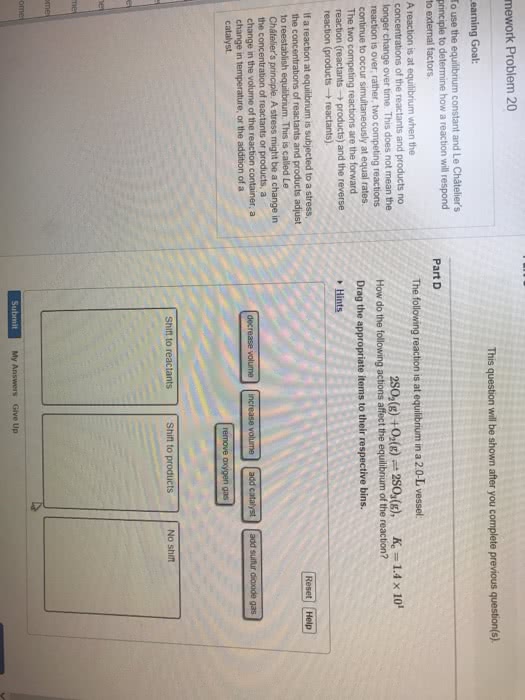
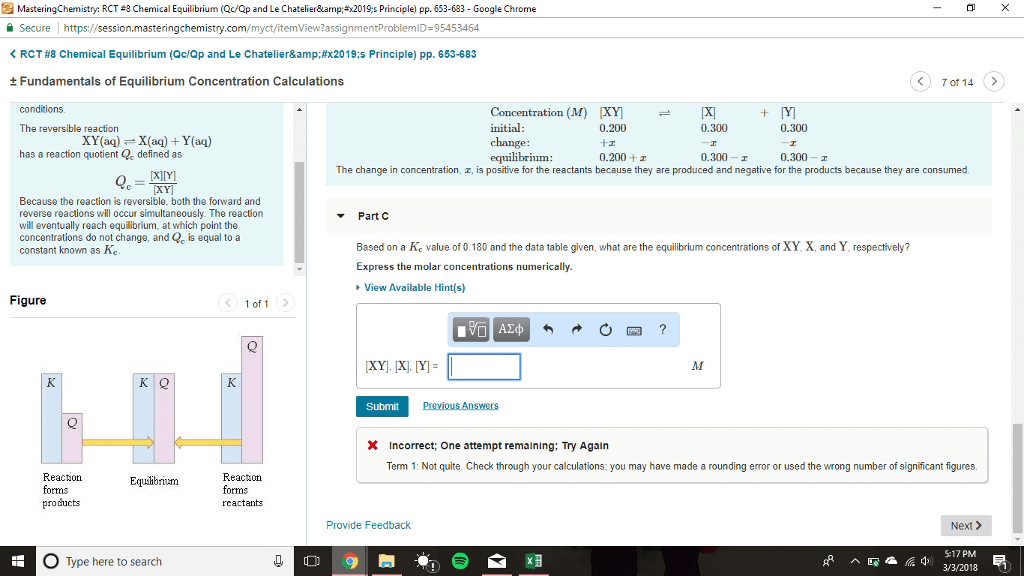
Chemical equilibrium part iii: if a condition is changed in a system, equilibrium adjusted to release stress, the reaction quotient q shifts upon changes in, concentration of one or more chemical species, pressure, temperature. Adding reactants/products: nreactants mproducts, q = [(cid:3043)(cid:3045)(cid:3042)(cid:3031)(cid:3048)(cid:3030)(cid:3047)(cid:3046)](cid:3288) Initial change equilibrium (ice) tables: consider n2 + 3h2 2nh3, knowing kc we can determine the final concentrations of products and reactants, if we know the initial concentrations, we always have the following proportionalities: 1 mol n2 is proportional to 2 mol nh3. 3 mol h2 is proportional to 2 mol nh3. -and : change only one number at a time and the rest comes out of proportionality. Ice tables, how to: consider a chemical reaction: Calculate the equilibrium concentrations of reactants and products when 0. 355 moles of cocl2 are introduced into a 1. 00 l vessel at 350k: fill in the table.