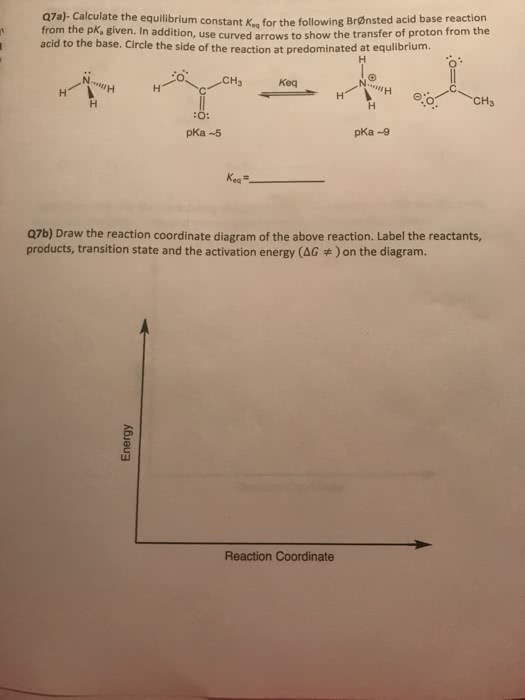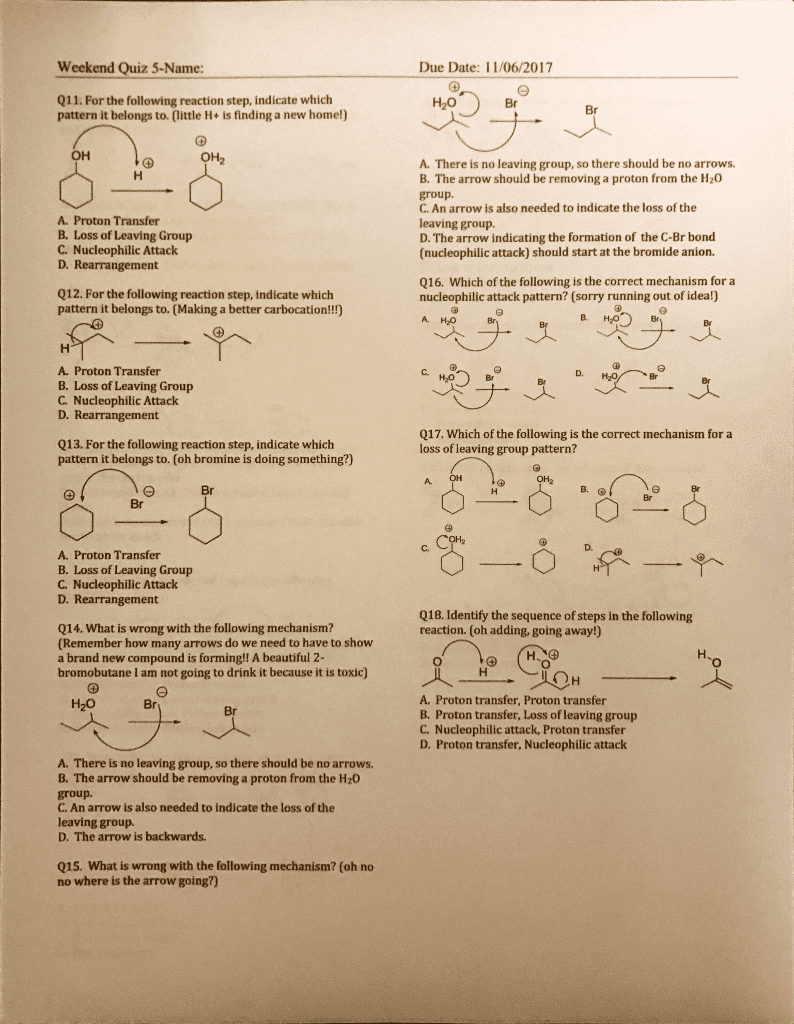
Weekend Quiz 5-Name Due Date: 11/06/2017 Q11. For the following reaction step, indicate which pattern it belongs to. (little H+ is finding a new home!) H2O Br OH A. There is no leaving group, so there should be no arrows. B. The arrow should be removing a proton from the H20 group A. Proton Transfer B. Loss of Leaving Group C. Nucleophilic Attack D. Rearrangement C. An arrow is also needed to indicate the loss of the leaving group. D. The arrow indicating the formation of the C-Br bond (nucleophilic attack) should start at the bromide anion. 012. For the following reaction step, indicate which pattern it belongs to. (Making a better carbocation!!) Q16. Which of the following is the correct mechanism for a nucleophilic attack pattern? (sorry running out of idea!) A. H2O A. Proton Transfer B. Loss of Leaving Group C Nucleophilic Attack D. Rearrangement H20 Q13. For the following reaction step, indicate which pattern it belongs to. (oh bromine is doing something?) Q17. Which of the following is the correct mechanism for a loss of leaving group pattern? Br A. Proton Transfer B. Loss of Leaving Group C. Nucleophilic Attaclk D. Rearrangement 018. Identify the sequence of steps in the following reaction. (oh adding, going away!) Q14. What is wrong with the following mechanism? (Remember how many arrows do we need to have to show a brand new compound is forming!! A beautiful 2- bromobutane I am not going to drink it because it is toxic) A. Proton transfer, Proton transfer B. Proton transfer, Loss of leaving group C. Nucleophilic attack, Proton transfer D. Proton transfer, Nucleophilic attack H20 Br A. There is no leaving group, so there should be no arrows. B. The arrow should be removing a proton from the H20 group. C. An arrow is also needed to indicate the loss of the leaving group. D. The arrow is backwards. Q15. What is wrong with the following mechanism? (oh no no where is the arrow going?)