CHEM 1032 Lecture Notes - Lecture 15: Equilibrium Constant
21 views3 pages
27 Feb 2017
School
Department
Course
Professor
Document Summary
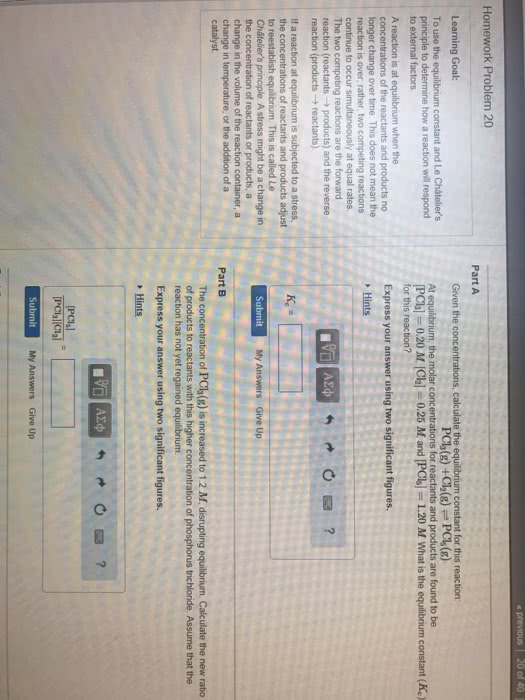
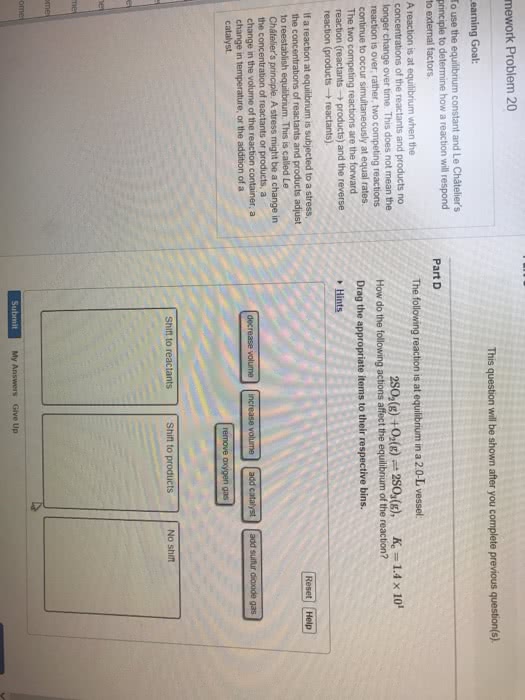
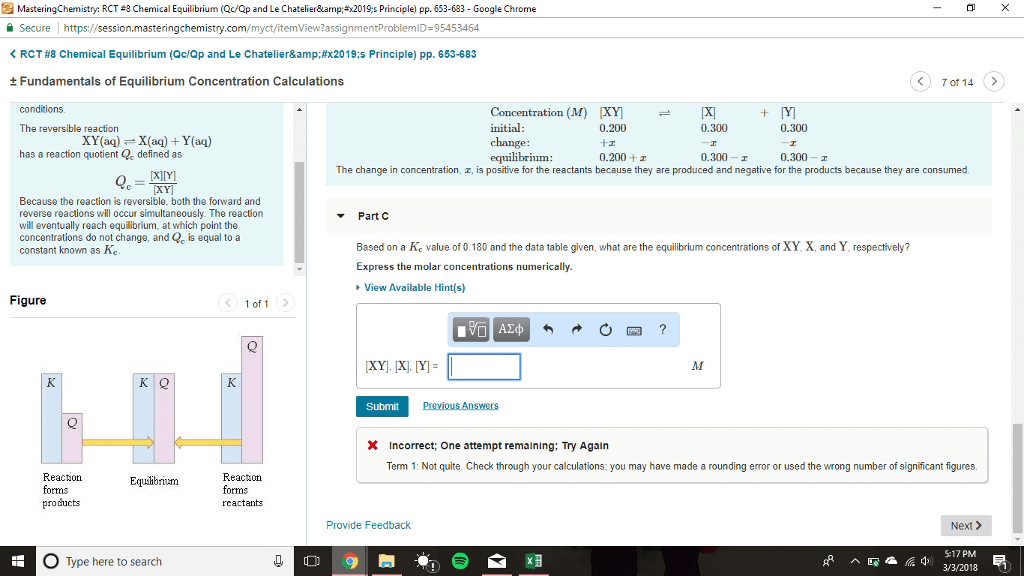
Equilibrium: rate forward = rate backward, concentrations of reactants and products are constant once at equilibrium, quantified with equilibrium constant (k) Concentrations at reestablished equilibrium will be different but k is the same. I can observe a similar thing if i add more reactants at equilibrium: forward rate > backwards rate, q < k. Le ch telier"s principle: when a chemical reaction at equilibrium is disturbed the system shifts in a direction that minimizes the stress, disturbances could be: Volume changes: constant temperature, for gases, i can think of volume changes in 2 ways: Volume decreases, concentration increases: if volume decreases, the reaction shifts to the side with less moles, if volume increases, the reaction shift to the side with more moles. Temperature change: if i change the temperature, i change the value of k, h + endothermic. Heat is the reactant: h - exothermic releasing heat. Heat is the product: heat + a + b c + d.
Get access
Grade+
$40 USD/m
Billed monthly

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
10 Verified Answers
Class+
$30 USD/m
Billed monthly

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
7 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232