01:160:162 Lecture Notes - Lecture 2: Partial Pressure, Chemical Formula, Molar Concentration
Document Summary
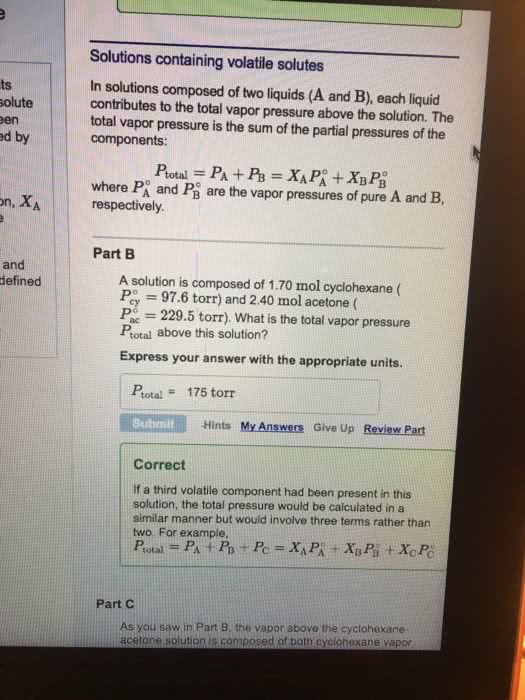
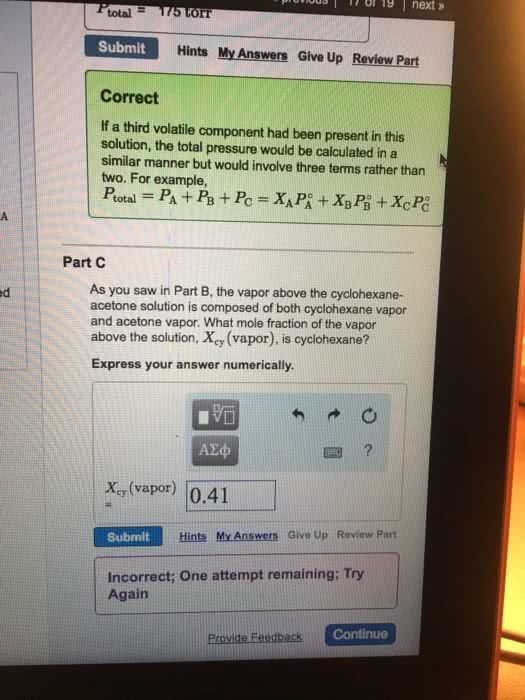
Vapor pressure of solutions vapor pressure of each component proportional to the mole fraction and the vapor pressure of the pure component. Assume the solution consists of solvent a and solute b, with mole fractions xa and xb. B where pa = vapor pressure of component a in the solution. P o xa = the mole fraction of a. The vapor pressure of the entire solution is the sum of pa+ pb. with corresponding definitions for component b. A solution is prepared by mixing 1. 00 mol of benzene and 1. 00 mol of toluene at 25. Pure toluene: vapor pressure = 28. 4 torr o. B = 28. 4 torr x b = 0. 50. Total vapor pressure = pa + pb = 47. 6 + 12. 2 = 61. 8 torr. Note that the vapor has a different composition than the liquid. The benzene (component a) started out as mole fraction 1/2 in the liquid, but more than half (0. 770) in the vapor.