CHEM 110 Lecture Notes - Lecture 9: Dinitrogen Tetroxide, Phosphorus Pentoxide, Iodine
Document Summary
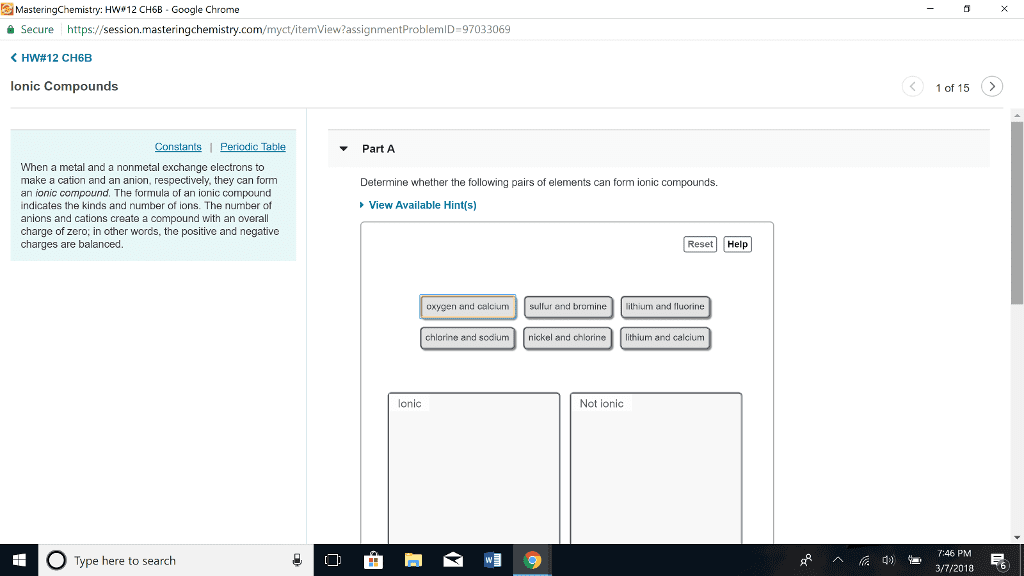
Writing formulas of ionic compounds from the identities of the component ions. Metals have a charge equal to group number. Nonmetals have a charge equal to the group number minus eight. Cations and anions must combine to give a formula with a net charge of zero. It must have the same number of positive charges as negative charges. Predict the formula of the ionic compounds formed from the combining ions of the following pairs of elements: Name the cation followed by the name of the anion. A positive ion retains the name of the element; change the anion suffix to -ide. If the cation of an element has several ions of different charges (as with transition metals) use a roman numeral following the metal name. Roman numbers give the charge of the metal. Use -ic to indicate the higher of the charges that ion might have. Use -ous to indicate the lower of the charges that ion might have.