CAS CH 102 Lecture Notes - Lecture 10: Alkyne, Homologous Series, Alkene
Document Summary
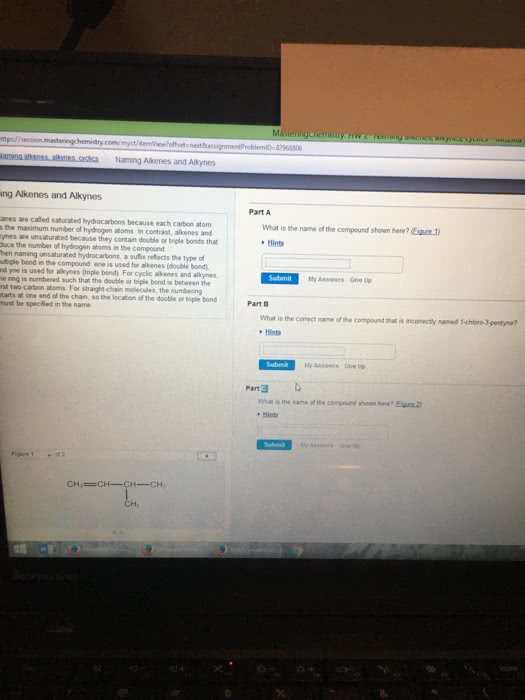
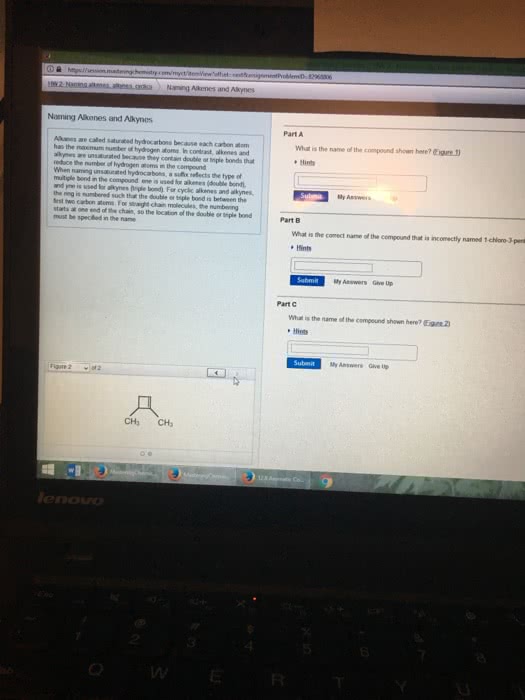
A hydrocarbon compound is a molecule consisting of carbon and hydrogen atoms, only. Table p can be used to figure out which prefixes denote the amount of carbons in a compound. Table q can be used to find out the number of hydrogens in each type of compound per a certain amount of carbons (homologous series: alkane, alkene, alkyne). Alkanes are named by using the prefix denoting the amount of carbons in the compound with the suffix -ane. They consist entirely of single bonds between carbons. Alkanes are the only hydrocarbon compounds that are saturated. Alkenes are named by using the prefix denoting the amount of carbons in the compound with the suffix -ene. Alkenes have only one double bond (carbon to carbon), the rest are single bonds. Alkynes are named by using the prefix denoting the amount of carbons in the compound with the suffix -yne.