CHEM 261 Lecture Notes - Lecture 6: Heat Capacity, Joule, Enthalpy
54 views3 pages
18 Oct 2016
School
Department
Course
Professor
Get access
Grade+
$40 USD/m
Billed monthly

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
10 Verified Answers
Class+
$30 USD/m
Billed monthly

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
7 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232
Related Documents
Related Questions
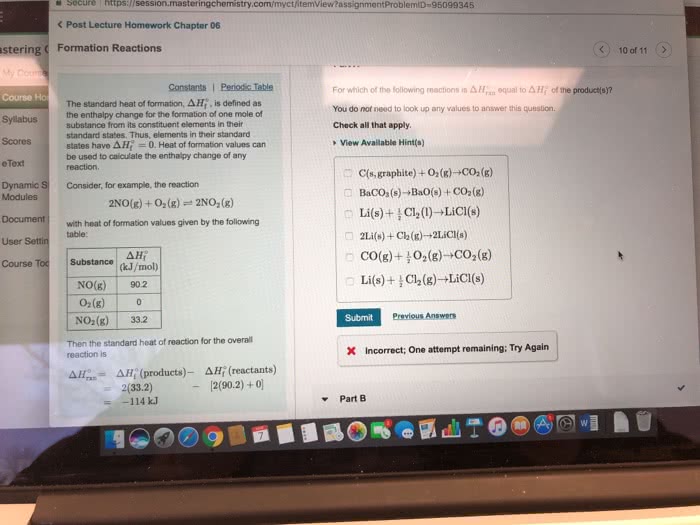
The standard enthalpy of formation (ÎHâf) is the enthalpy change that occurs when exactly 1 mol of a compound is formed from its constituent elements under standard conditions. The standard conditions are 1 atm pressure, a temperature of 25 âC , and all the species present at a concentration of 1 M . A "standard enthalpies of formation table" containing ÎHâf values might look something like this:
| Substance | ÎHâf |
| H(g) | 218 kJ/mol |
| H2(g) | 0 kJ/mol |
| Sr(s) | 0 kJ/mol |
| Sr2+(aq) | â 549.7 kJ/mol |
| C(g) | 71 kJ/mol |
| C(s) | 0 kJ/mol |
| N(g) | 473 kJ/mol |
| O2(g) | 0 kJ/mol |
| O(g) | 249 kJ/mol |
| S2(g) | 129 kJ/mol |
What is the balanced chemical equation for the reaction used to calculate ÎHâf of SrCO3(s)?