CHEM 120 Lecture Notes - Lecture 7: Kinetic Theory Of Gases, Collision Theory, Activated Complex
46 views4 pages
10 Feb 2016
School
Department
Course
Professor

26
CHEM 120 Full Course Notes
Verified Note
26 documents
Document Summary
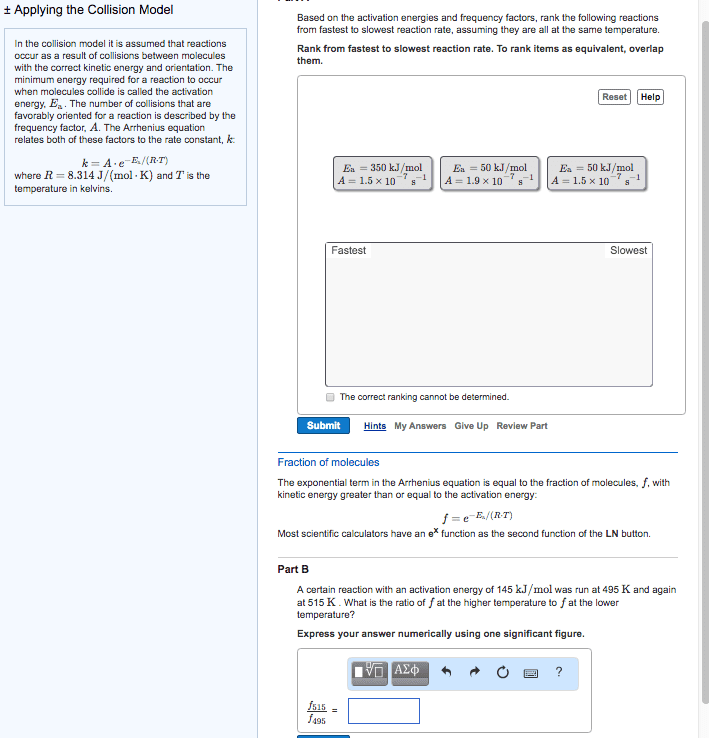
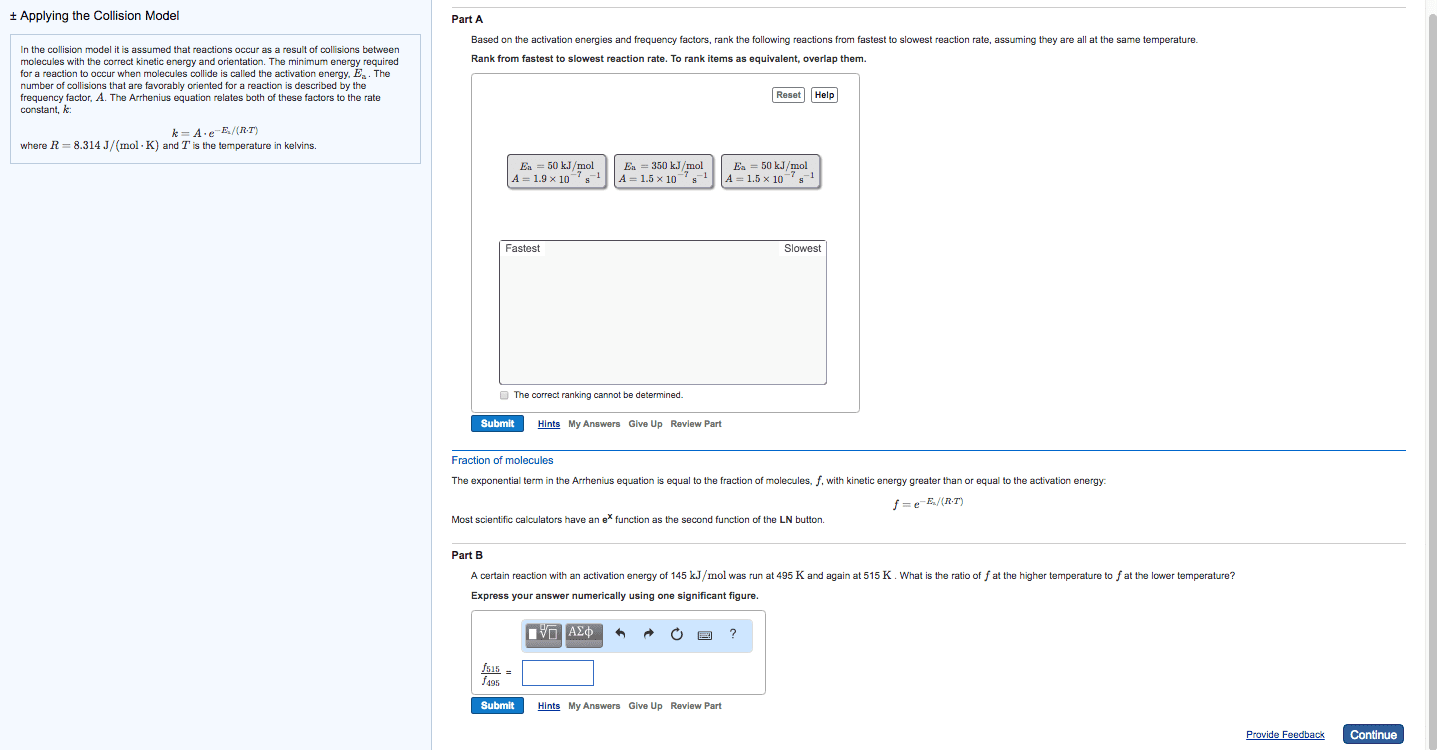
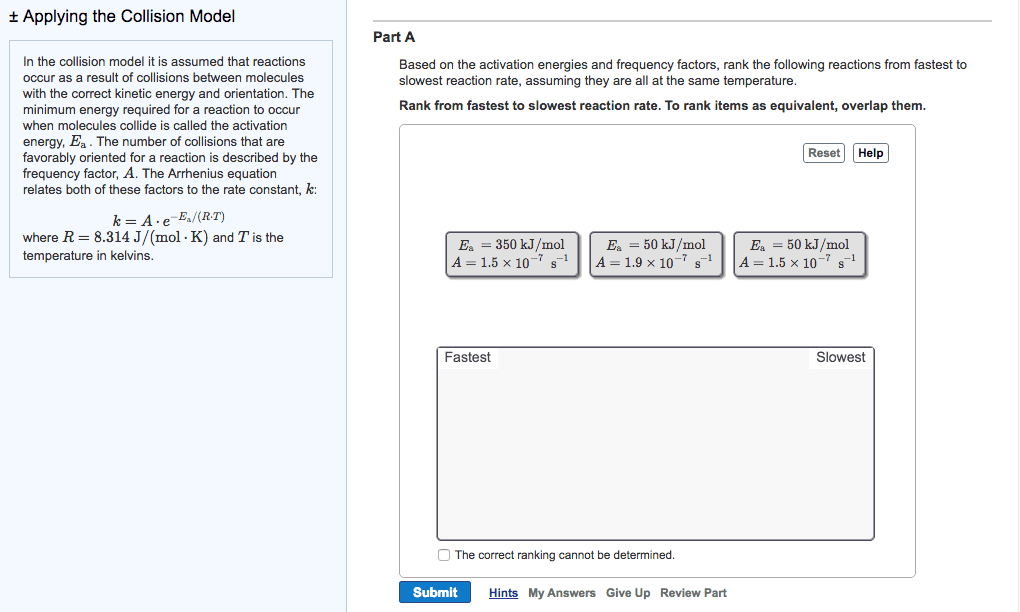
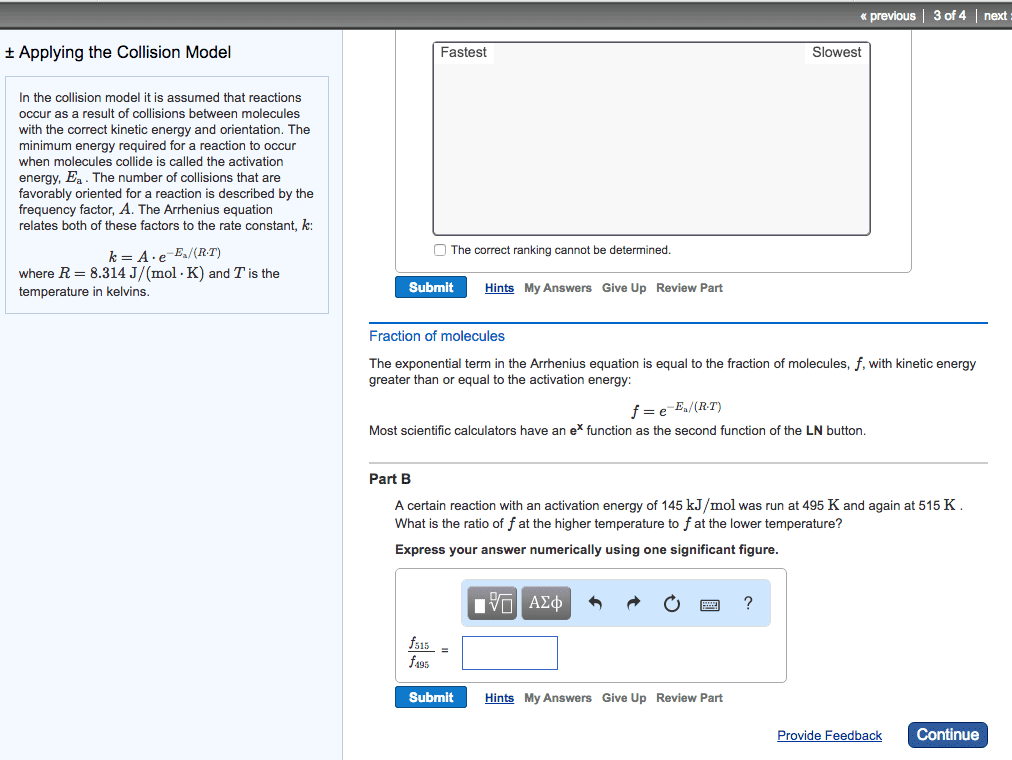
Chem 120 lecture 7 collision theory & reaction mechanisms. 14. 8: theoretical models for chemical kinetics: collision theory. Kinetic- molecular theory can be used to calculate the collision frequency of a gas: in gases at stp, ~10^30 collisions occur per second in 1l! huge! If each reaction proceeded at that rate/if each collision produced a reaction, then the rate would be huge! ~10^30/na: actual rates in gas reactions are generally much slower a typical value being. 10^-4m/s thus only a small fraction of collisions produce a reaction. Not all collisions result in a chemical reaction (ie. only collisions between molecules whose combines ke is above the minimum will react) For a reaction to happen, there must be a redistribution of energy sufficient to. Activation energy (ea) = the minimum ke that molecules need to bring to their collisions for a chemical reaction to occur. Kinetic energy distribution: (only some particles react. Shaded area is proportional to e-ea/rt, therefore dependent on temperature.
Get access
Grade+
$40 USD/m
Billed monthly

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
10 Verified Answers
Class+
$30 USD/m
Billed monthly

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
7 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232