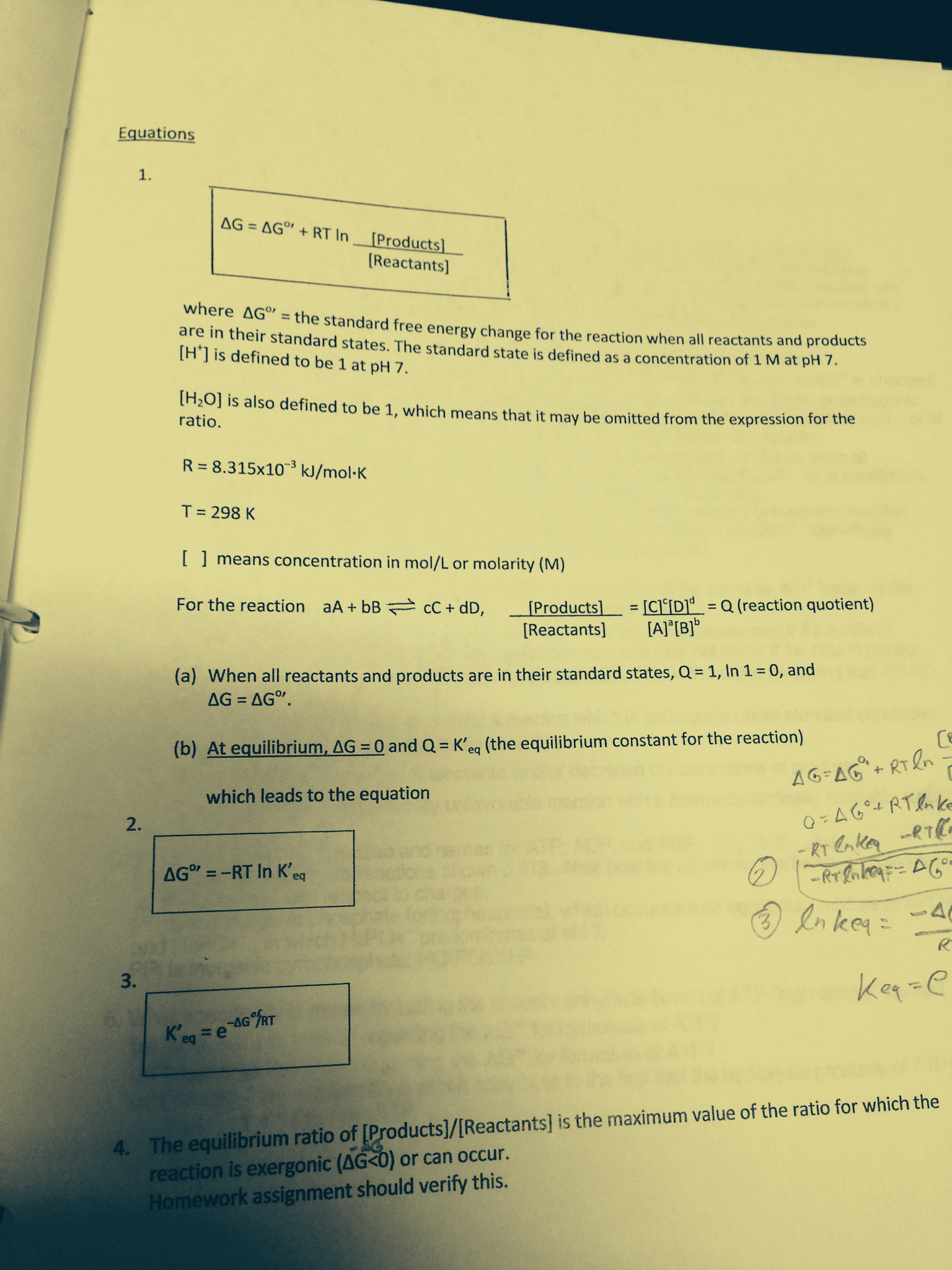Please answer the remaining blanks in the table and thequestions underneath the table to receive fullcredit. Please show work.


State your conclusions from the above data by underlining one of the two choices in each parentheses. DeltaG is negative for all values of [Products]/[Reactants] which are (less than or greater than) K'eq, meaning that these are the conditions under which the reaction (can or cannot) occur. An endergonic reaction can be changed to an exergonic reaction by increasing the (reactants or products) and/or by decreasing the (reactants or products). Exergonic under standard conditions or equiulibrium favors products (K' eq > 1) where Delta G degree' = the standard free energy change for the reaction when all reactants and products are states. The standard state is defined as a concentration of 1 M at pH 7. [H+] is defined to be 1 at pH 7. [H2O] is also defined to be 1, which means that it may be omitted from the expression for the ratio. R = 8.315 times 10-3 kJ/mol-K T = 298 K [ ] means concentration in mol/L or molarity (M) For the reaction aA + bB cC + dD, [Products] / [Reactants] = [C]c[D]d = Q. (reaction quotient) When all reactants and products are in their standard states, 0.= 1, In 1 = 0, and Delta G = Delta G degree'. At equilibrium, Delta G = 0 and Q = K'eq (the equilibrium constant for the reaction) which leads to the equation The equilibrium ratio of [Products]/[Reactants] is the maximum value of the ratio for which the reaction is exergonic (Delta G
Show transcribed image text State your conclusions from the above data by underlining one of the two choices in each parentheses. DeltaG is negative for all values of [Products]/[Reactants] which are (less than or greater than) K'eq, meaning that these are the conditions under which the reaction (can or cannot) occur. An endergonic reaction can be changed to an exergonic reaction by increasing the (reactants or products) and/or by decreasing the (reactants or products). Exergonic under standard conditions or equiulibrium favors products (K' eq > 1) where Delta G degree' = the standard free energy change for the reaction when all reactants and products are states. The standard state is defined as a concentration of 1 M at pH 7. [H+] is defined to be 1 at pH 7. [H2O] is also defined to be 1, which means that it may be omitted from the expression for the ratio. R = 8.315 times 10-3 kJ/mol-K T = 298 K [ ] means concentration in mol/L or molarity (M) For the reaction aA + bB cC + dD, [Products] / [Reactants] = [C]c[D]d = Q. (reaction quotient) When all reactants and products are in their standard states, 0.= 1, In 1 = 0, and Delta G = Delta G degree'. At equilibrium, Delta G = 0 and Q = K'eq (the equilibrium constant for the reaction) which leads to the equation The equilibrium ratio of [Products]/[Reactants] is the maximum value of the ratio for which the reaction is exergonic (Delta G