CHEM101 Lecture Notes - Lecture 14: Rate Equation, Reaction Rate, Burette
Document Summary
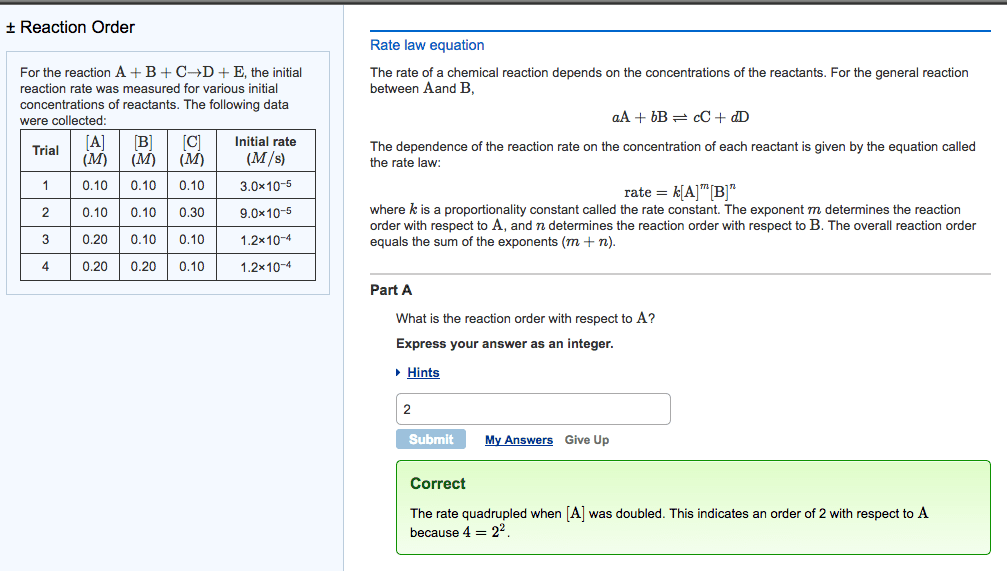
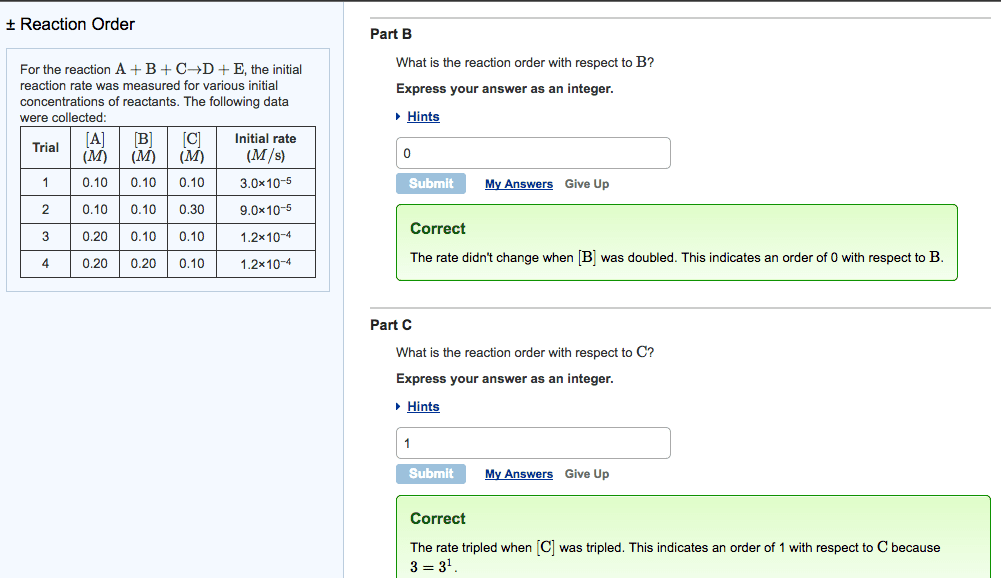
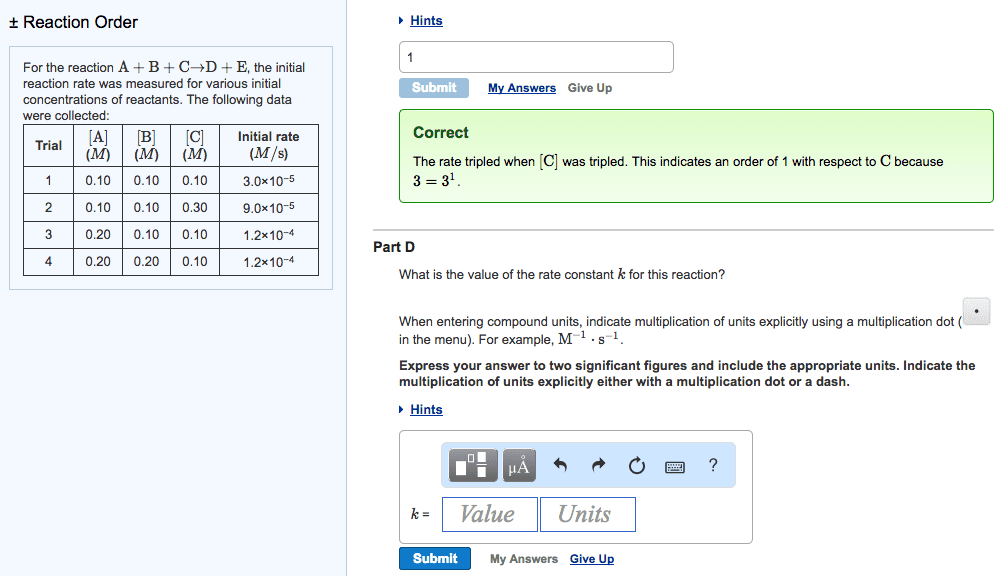
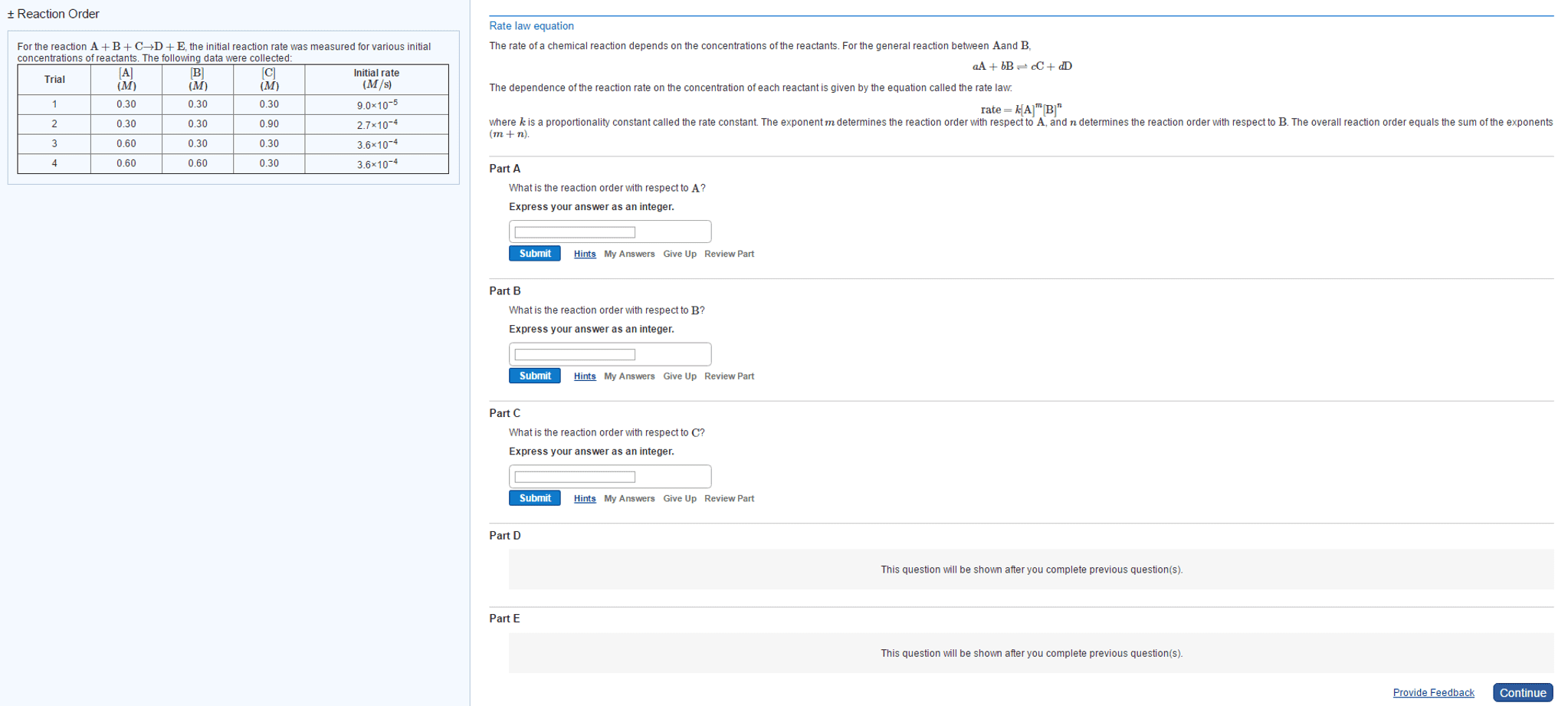
Stoichiometry: the rate of disappearance of reactants must equal the rate of appearance of products in the correct stoichiometric ratio. Concentration and rate: one can gain information about the rate of a reaction and hence the mechanism of the reaction by seeing how the rate changes with changes in concentration of reactants, e. g. doubled concentration = doubled concentration. Rate law: k = [a]m [b]n, shows how the rate depends on the concentration of reactants, order of reaction is measured experimentally, to solve k, k = rate. [a]m [b]n: practice problem see lecture notes. Zero order: to determine a concentration at a given time: First order: to determine a concentration at a given time: Second order: to determine a concentration at a given time: Measuring rates: a reaction rate is based on change over time, can be measured via, the disappearance of a product of a reactant or the appearance of a product.