CHEM10006 Lecture Notes - Lecture 31: Covalent Bond, Lone Pair, Nernst Equation
19 views2 pages
10 Aug 2018
School
Department
Course
Professor
Document Summary
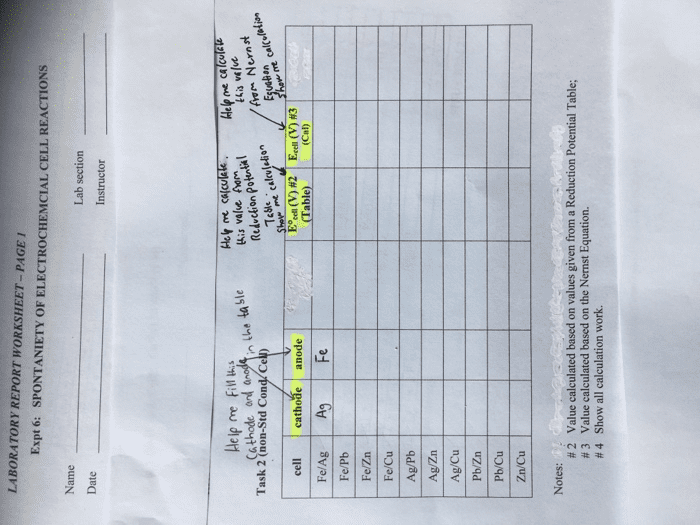
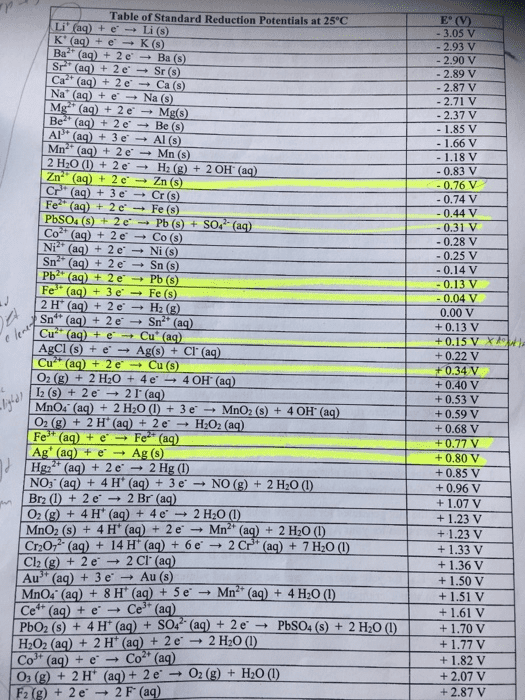
The ne(cid:396)(cid:374)st e(cid:395)uatio(cid:374) allows us to calculate the pote(cid:374)tial at i(cid:374)te(cid:396)(cid:373)ediate ph"s. Standard reduction potentials depend on ph, and are based on unit concentrations. As ph = - log[h+], ecell = eo - 0. 0591 ph, which will graph to a straight line. Plot of e vs ph for the reduction of o2 and h2o. The stability field of water is the range of values of potential and ph for which water is thermodynamically stable towards both oxidation and reduction. Species with a potential more positive than the top line will release o2 from water. Species with a potential more negative than the bottom line will release h2 from water. Lines mark places where two species exist in equilibrium. Pure redox reactions are horizontal lines - these reactions are not ph-dependent. Pure acid-base reactions are vertical lines - these do not depend on potential. Reactions that are both acid-base and redox have a slope of -0. 0592 v/ph.
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232