CBMS107 Lecture Notes - Lecture 12: Enantiomer, Stereocenter, Optical Rotation
Document Summary
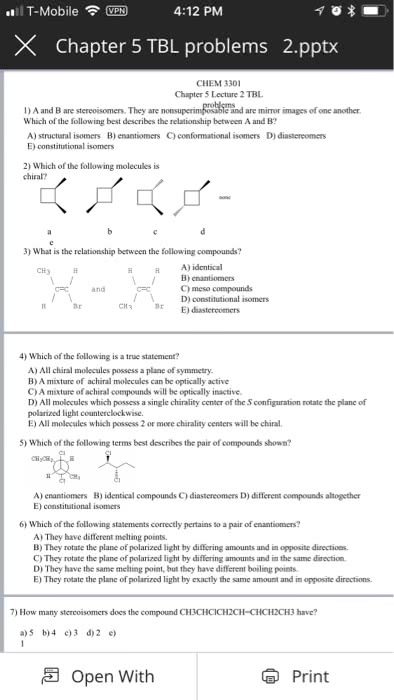
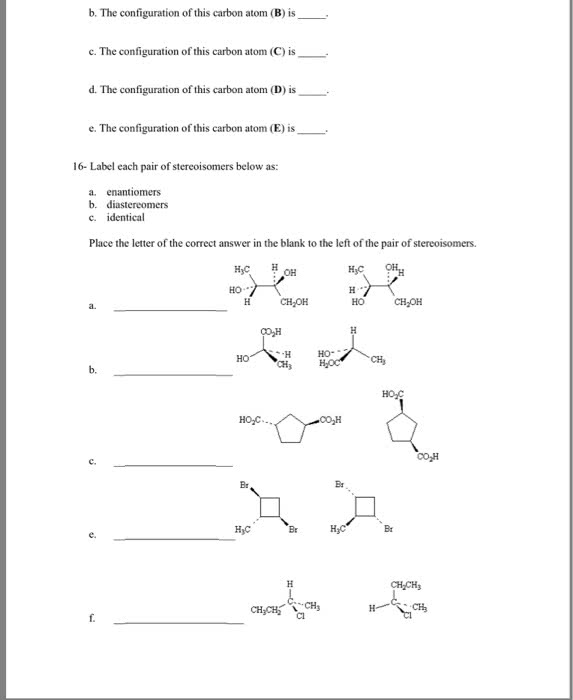
3rd 4th april 2018: lecture 16 and 17. Compounds whose atoms are connected differently (different carbon skeleton: stereoisomers: Isomers with the same order of attachment of atoms, but a different orientation of their atoms or groups of atoms in space. Stereoisomers whose molecules are mirror images of each other. Stereoisomers whose molecules are not mirror images of each other. Examples include: geometric isomers (arising from restricted rotation: cis/trans isomers of cycloalkanes (due to restricted rotation of ring bonds, cis/trans isomers of alkenes (due to restricted rotation of a double bond, chirality: Objects whose mirror image is not superimposable (they cannot be placed on top of each other) upon each other are said to be chiral. Occurs if any of the tetrahedral carbon atoms in the molecules have 4 different groups attached. Purpose of chirality: for example, a pair of enantiomers, one fits into the protein receptors to give a biological response, the other does not.