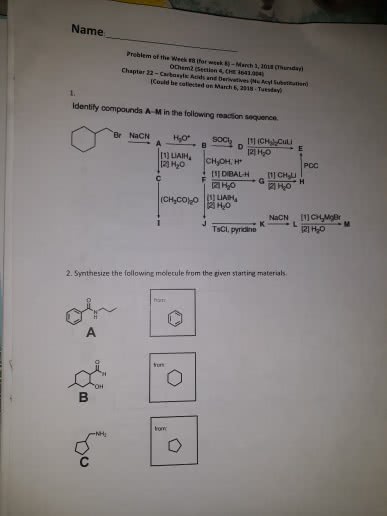
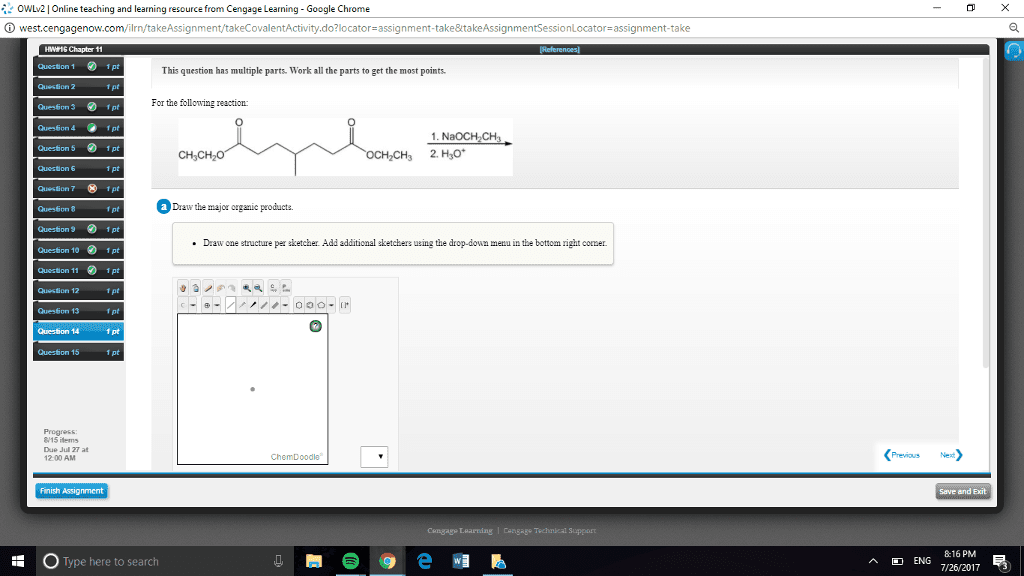
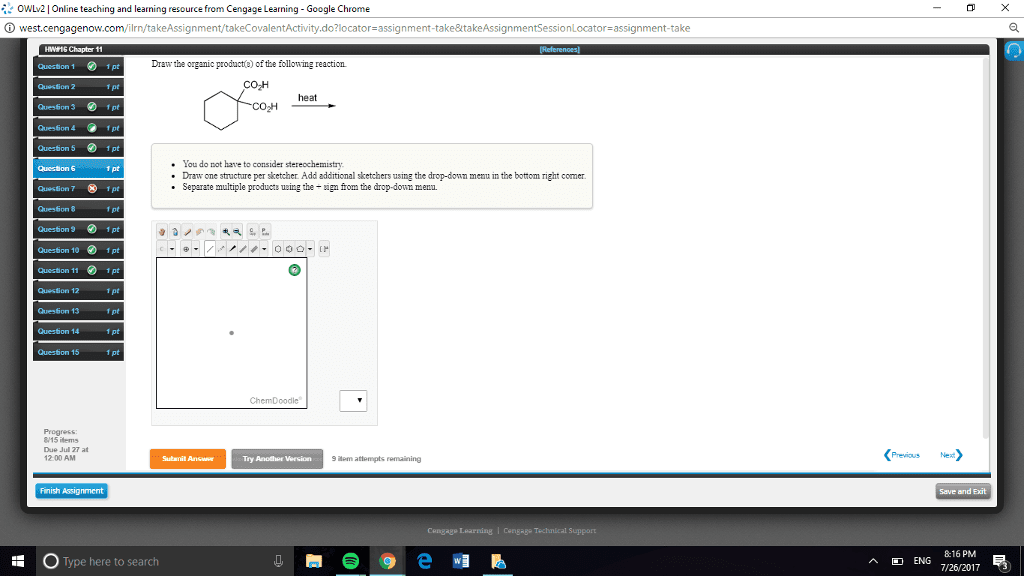
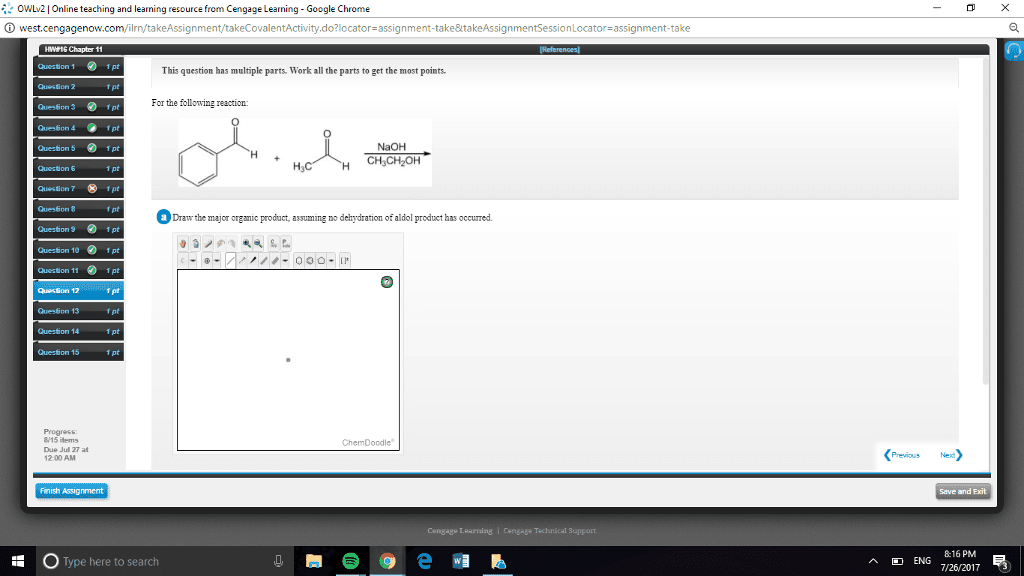
CHEN3010 Lecture Notes - Lecture 10: Pearson Education, Mental Chronometry, Reaction Rate Constant

Chemical Reaction Engineering
Unit CHEN3010/5040
Semester 1 / 2018
Week 6
Lecture 1
Multiple Reaction
(Fogler Chapter 8)
Gia Hung Pham
find more resources at oneclass.com
find more resources at oneclass.com

Objectives
•Discuss general mole balances, net rates, and relative rates for
multiple reactions.
•Define the selectivity parameter for multiple reactions.
•Select a suitable reactor for multiple reactions.
•Modify the CRE algorithm to solve reaction engineering problems of
multiple reactions.
find more resources at oneclass.com
find more resources at oneclass.com

Multiple Reaction
Four chemical reaction types:
- Parallel
- Series
- Independent
- Complex
find more resources at oneclass.com
find more resources at oneclass.com
Document Summary
Reactant is consumed by two different reaction pathways to form different products. Reactant is consumed to form products b, then product b is consumed to form product c. one of these products can be an undesired product. Multiple reactions that involve a combination of both series and parallel reactions. Neither products nor reactants react with each other. In this chapter, we learn how to design reactor as well as the best configuration for optimizing product formation for different types of multiple reaction systems in practice. We try to maximise d and minimise u. Selectivity tells us how one product is favoured over another. Number of mole of d at the end of the reaction. Number of mole of u at the end of the reaction. Overall yield (ratio of moles product to number of moles of key reactant a consumed) Maximizing the desired product / reactor selection and operating conditions.