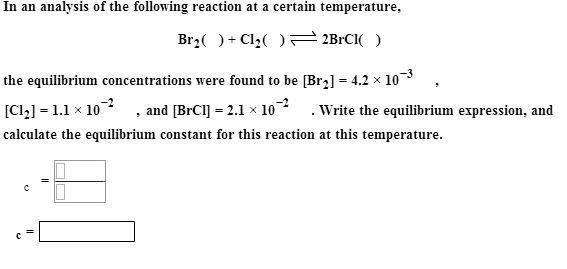Show transcribed data
13. A solution contains 3.25 x  hydroxide ions. What is the pH of the solution?
hydroxide ions. What is the pH of the solution?
A. 3.25
B. 6,51
C. 7.49
D. 8,00
E. none of the above
14. Identify the weakest acid,
A. HCN  = 62 x
= 62 x 
B. HCOOH  = 1.8 x
= 1.8 x 
C. 
 = 72 x
= 72 x 
D. HF  = 66 x
= 66 x 
E. All acids are the same strength, as long as they have a dissociable hydrogen ion.
15. The equilibrium system shown below was analyzed and the concentrations of 
 and
and  were found, in mol/L, to be 4.4, 3.2, 1.5 respectively. The equilibrium constant must be which of the following?
were found, in mol/L, to be 4.4, 3.2, 1.5 respectively. The equilibrium constant must be which of the following?

A. 4.0
B. 0.25
C. 0.92
D. 1.1
E. 0.16
16. The equilibrium shown below was analyzed and the concentrations of  ,
,  and
and  were found in mol/L, to be 4.5, 2.6 and 12. The equilibrium constant must be which of the following?
were found in mol/L, to be 4.5, 2.6 and 12. The equilibrium constant must be which of the following?

A. 1.4
B. 0.69
C. 9.8
D. 0.84
E. 1.2,
17. A full syringe contains this equilibrium

If nitrogen dioxide gas is brown and the other gas is colourless, what is observed when the syringe is used to compress this gas mixture?
A. the mixture gets darker
B. the mixture gets lighter
C. no change in colour of the mixture
D. the mixture is colourless
18. Consider this equilibrium

If some nitrogen gas were injected into this system at constant temperature and pressure the system would most likely experience which of the following?
A. an increase in (hydrogen)
B. a decrease in (ammonial)
C. a decrease in (hydrogen)
D. an increase in (ammonia)
E. both c and d
19. Consider the following system at equilibrium

Which of the following actions would cause the  to increase?
to increase?
I. increasing the volume at constant pressure and temperature
II. decreasing the volume at constant pressure and temperature
III. increasing the temperature
IV. adding bromine gas at constant temperature and pressure
V. removing ethyne gas at constant temperature and pressure
VI. decreasing the temperature
A. I and II
B. III and VI
C. IV and V
D. III
E. VI
Show transcribed data
13. A solution contains 3.25 x hydroxide ions. What is the pH of the solution?
A. 3.25
B. 6,51
C. 7.49
D. 8,00
E. none of the above
14. Identify the weakest acid,
A. HCN = 62 x
B. HCOOH = 1.8 x
C.
= 72 x
D. HF = 66 x
E. All acids are the same strength, as long as they have a dissociable hydrogen ion.
15. The equilibrium system shown below was analyzed and the concentrations of
and
were found, in mol/L, to be 4.4, 3.2, 1.5 respectively. The equilibrium constant must be which of the following?
A. 4.0
B. 0.25
C. 0.92
D. 1.1
E. 0.16
16. The equilibrium shown below was analyzed and the concentrations of ,
and
were found in mol/L, to be 4.5, 2.6 and 12. The equilibrium constant must be which of the following?
A. 1.4
B. 0.69
C. 9.8
D. 0.84
E. 1.2,
17. A full syringe contains this equilibrium
If nitrogen dioxide gas is brown and the other gas is colourless, what is observed when the syringe is used to compress this gas mixture?
A. the mixture gets darker
B. the mixture gets lighter
C. no change in colour of the mixture
D. the mixture is colourless
18. Consider this equilibrium
If some nitrogen gas were injected into this system at constant temperature and pressure the system would most likely experience which of the following?
A. an increase in (hydrogen)
B. a decrease in (ammonial)
C. a decrease in (hydrogen)
D. an increase in (ammonia)
E. both c and d
19. Consider the following system at equilibrium
Which of the following actions would cause the to increase?
A. I and II
B. III and VI
C. IV and V
D. III
E. VI