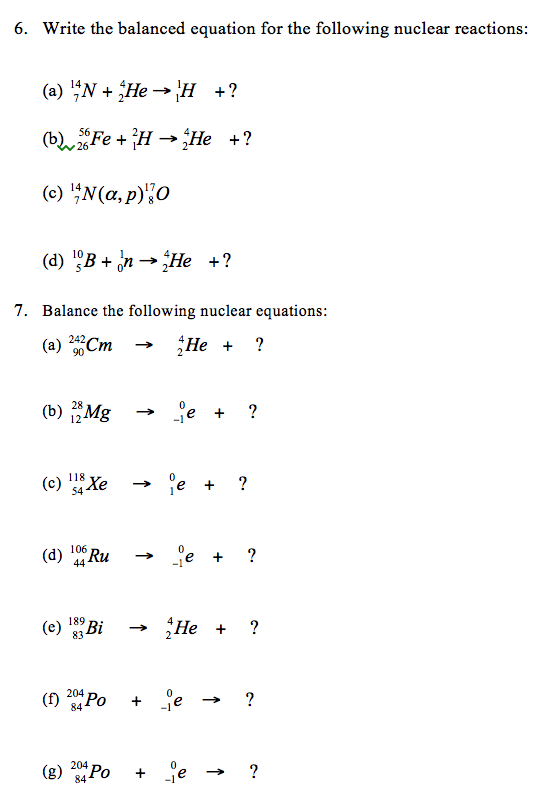2
answers
1
watching
84
views
anyaforgerLv1
3 Jul 2022
Show transcribed data
1) The isomerization reaction shown below follows first order kinetics at 500 K.
 a) Use the rate constant you determined in example 3 on page 4 to determine the half life of this reaction.
b) Use the half life you just determined to predit how long it will take for the concentration of
a) Use the rate constant you determined in example 3 on page 4 to determine the half life of this reaction.
b) Use the half life you just determined to predit how long it will take for the concentration of  to reach 25% of its original value?
c) Compare the answer you got here to the one you got in example 3 and discuss whether the two answers are in agreement. Explain why the answers are/are not in agreement.
Compare the two methods and determine under what circumstances you can use which method.
2) The radioactive decay of 14C is a first order process with a half life of 5730 years. If the living wood gives 15.3 disintegrations per minute per gram, and a wooden bowl found in an archeological dig gives 6.29 disintegrations per minute per gram, how old is the bowl?
3) Ba-141 has a half life of 18.26 minutes and decays by first order kinetics. How many half lives will it take for a mole of Ba-141 to decay down to one atom?
4) 1-131 decays by first order kinetics. What will be the concentration of J-131 after 65 half lives if you start with a 2.57 x
to reach 25% of its original value?
c) Compare the answer you got here to the one you got in example 3 and discuss whether the two answers are in agreement. Explain why the answers are/are not in agreement.
Compare the two methods and determine under what circumstances you can use which method.
2) The radioactive decay of 14C is a first order process with a half life of 5730 years. If the living wood gives 15.3 disintegrations per minute per gram, and a wooden bowl found in an archeological dig gives 6.29 disintegrations per minute per gram, how old is the bowl?
3) Ba-141 has a half life of 18.26 minutes and decays by first order kinetics. How many half lives will it take for a mole of Ba-141 to decay down to one atom?
4) 1-131 decays by first order kinetics. What will be the concentration of J-131 after 65 half lives if you start with a 2.57 x  grams of I-131.
grams of I-131.
Show transcribed data
1) The isomerization reaction shown below follows first order kinetics at 500 K.
a) Use the rate constant you determined in example 3 on page 4 to determine the half life of this reaction.
b) Use the half life you just determined to predit how long it will take for the concentration of  to reach 25% of its original value?
to reach 25% of its original value?
c) Compare the answer you got here to the one you got in example 3 and discuss whether the two answers are in agreement. Explain why the answers are/are not in agreement.
Compare the two methods and determine under what circumstances you can use which method.
2) The radioactive decay of 14C is a first order process with a half life of 5730 years. If the living wood gives 15.3 disintegrations per minute per gram, and a wooden bowl found in an archeological dig gives 6.29 disintegrations per minute per gram, how old is the bowl?
3) Ba-141 has a half life of 18.26 minutes and decays by first order kinetics. How many half lives will it take for a mole of Ba-141 to decay down to one atom?
4) 1-131 decays by first order kinetics. What will be the concentration of J-131 after 65 half lives if you start with a 2.57 x  grams of I-131.
grams of I-131.
christopherc63Lv10
10 Nov 2022