1
answer
0
watching
116
views
12 Dec 2019
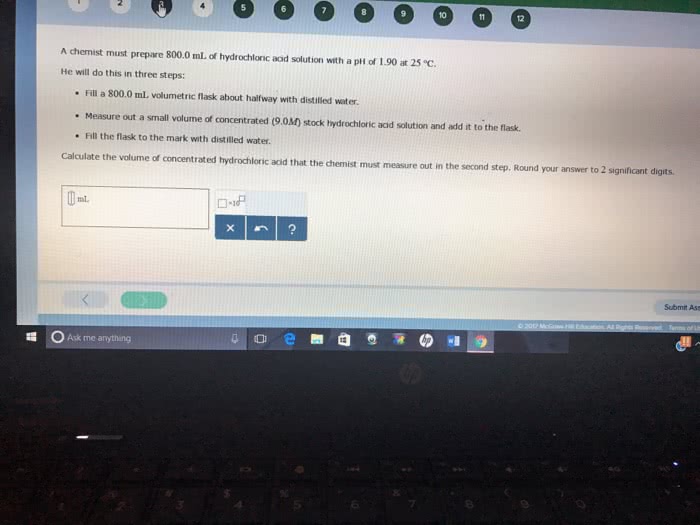
You wish to make a 0.445 M hydrochloric acid solution from a stock solution of 12.0 M hydrochloric acid. How much concentrated acid must you add to obtain a total volume of 150 mL of the dilute solution?
You wish to make a 0.445 M hydrochloric acid solution from a stock solution of 12.0 M hydrochloric acid. How much concentrated acid must you add to obtain a total volume of 150 mL of the dilute solution?
Lelia LubowitzLv2
13 Dec 2019