1
answer
0
watching
128
views
11 Dec 2019
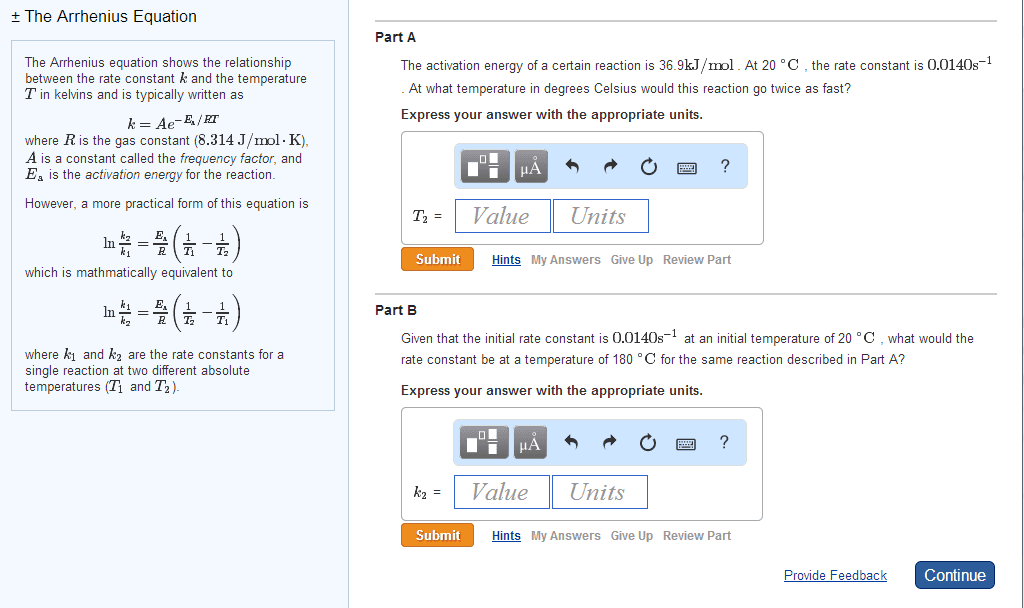
The activation energy of a certain reaction is 37.6 kJ/mol . At 21 âC , the rate constant is 0.0190s^-1 . At what temperature in degrees Celsius would this reaction go twice as fast?
Please, use Arrchenius eqn. : lnk2/k1=Ea/R (1/T1-1/T2) We are looking for T2? , but how do I know what is k2 when only k1 is given as 0.0190s^-1
The activation energy of a certain reaction is 37.6 kJ/mol . At 21 âC , the rate constant is 0.0190s^-1 . At what temperature in degrees Celsius would this reaction go twice as fast?
Please, use Arrchenius eqn. :
lnk2/k1=Ea/R (1/T1-1/T2)
We are looking for T2? , but how do I know what is k2 when only k1 is given as 0.0190s^-1
Beverley SmithLv2
13 Dec 2019