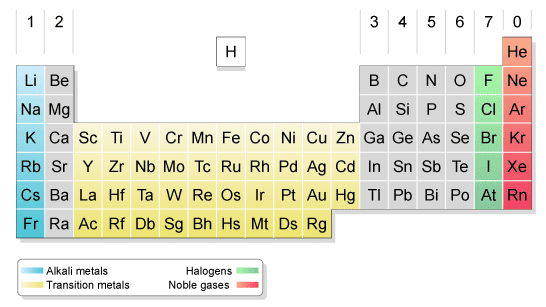
Calculate the number of protons, neutrons, and electrons in: 17 Cl; 11 Na; 36 Kr. The element lithium has two naturally occurring isotopes. One of these has a mass of 6.0151 amu and a natural abundance of 7.49%. A second isotope has a mass of 7.0160 amu and a natural abundance of 92.51%. Calculate the atomic mass of lithium. Which group of the periodic table is known as the alkali metals? List them. Which group of the periodic table is known as the halogens? List them. Which group of the periodic table is known as the noble gases? List them. For each of the elements Ca, K, Cu, Zn, Br, and Kr, provide the following information: Which are metals? Which are main-group metals? Which are inert or noble gases? Using the periodic table, write the electron configuration and orbital diagram of each of the following atoms: B; S; Ar. Which of the following pairs of atoms and/or ions are isoelectronic with one another? F-, Cl- K+, Ar Arrange each of the following lists of elements in order of increasing atomic size: N, O, F Li, K, Cs Cl, Br, I