CHEM 112 Lecture Notes - Lecture 4: Rate Equation, Arrhenius Equation, Reaction Rate
Document Summary
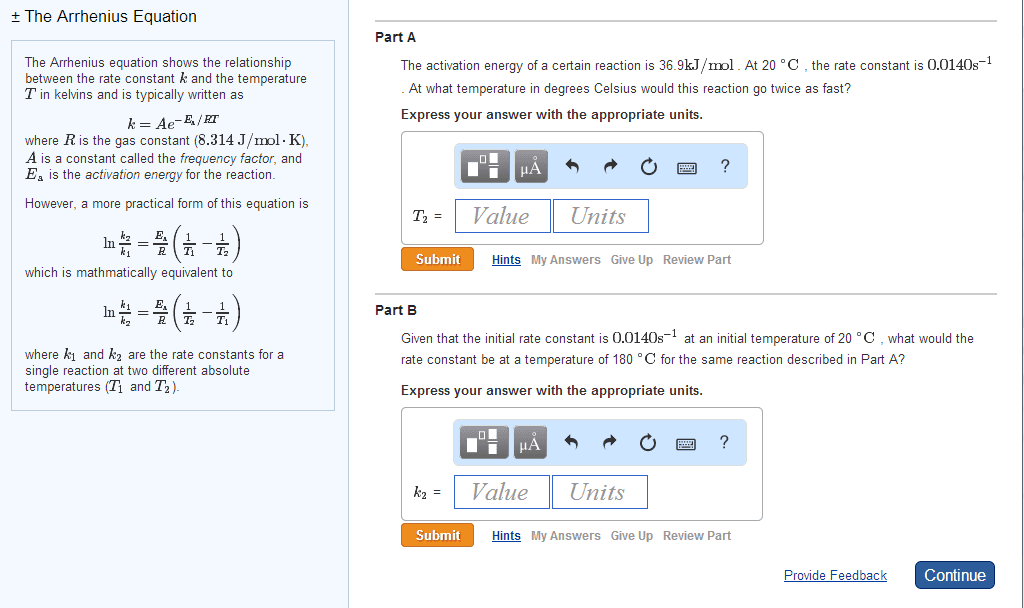
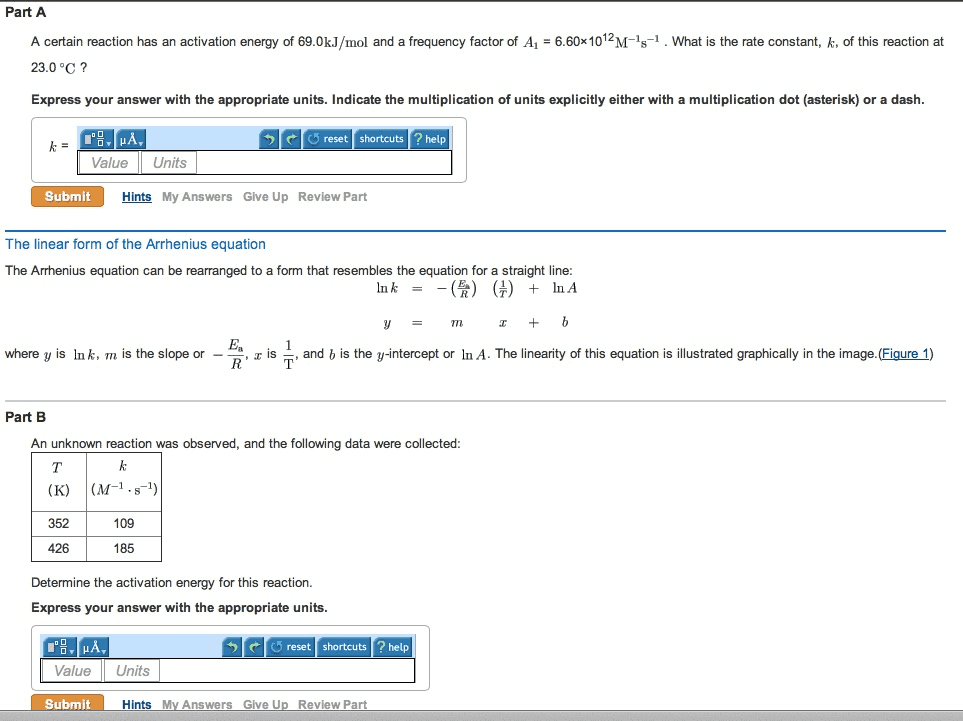
Remember, e and reaction rate are independent of each other. In slope-intercept formula: y=mx+b ln(k) = (-ea/r)(1/t) + ln(a) Deriving the formula if there is more than one k value (rate constant): ln(k1) ln(k2) = (-ea/r)(1/t) + ln(a: ln(k1/k2) = (-ea/r)(1/t2 1/t1) ln(k1/k2) = (ea/r)(1/t2 1/t1) Remember, the arrhenius equation combines 3 factors: collision frequency, orientation factor, fraction of molecules above the energy threshold (e ea) The activation energy is typically about half as strong as an atomic bond (~100 kj/mol vs ~200 kj/mol) Reaction concentration varies as a function of time. Rate = [x]/ t (m/s: instantaneous, average, and initial rate. Can be expressed in terms of changes of concentration of each reactant and product as a function of time; A + b = c + (d), where the reactants are a and b, the products are c and d, and , , and.