CHEM 110 Lecture Notes - Lecture 1: Atomic Orbital, Relative Atomic Mass, Atomic Number
46 views1 pages
Document Summary
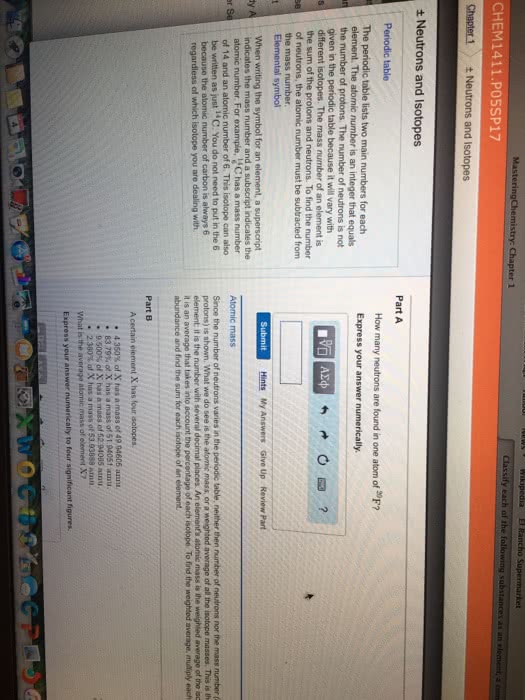
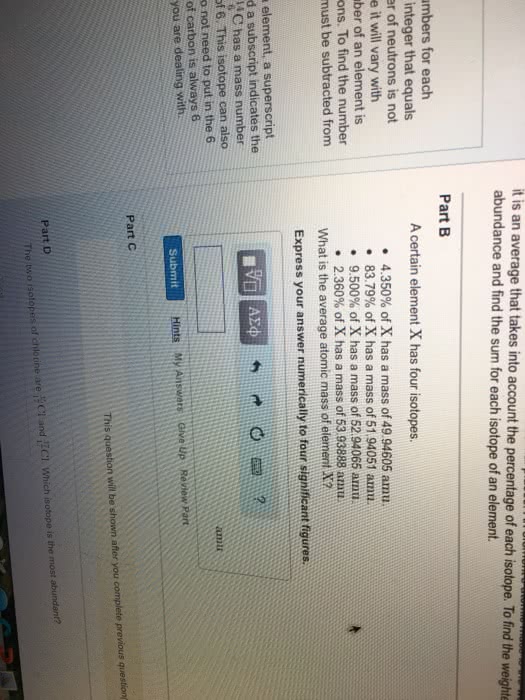
Coulomb"s law is the relationship that the electrons have to the protons. Electrons have a negative charge of -1. 602 x 10-19 coulmbs (c). An element with a different number of neutrons is known as an isotope. Atomic weight is the average of the masses of all isotopes. Only the electron cloud is modified in chemical reactions. 1 mol= the number of grams in atomic mass. Only the noble gasses exist as individuals (he, ne, ar, kr, xe, rn) Protons and neutron arrangements only change during nuclear reactions. Ions with opposite charges form ionic solids (salts) Allotropes- one of two or more forms of an element in the same phase. Molecular compounds- molecules made of more than one type of atom.
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232